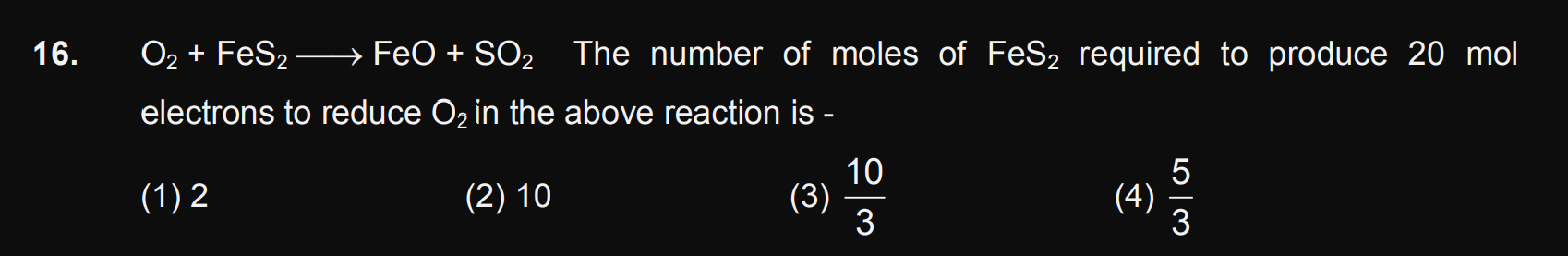JEE Class main Answered
What will be the molality of the solution containing 18.25 g of HCl gas in 500 g of water?
Asked by sayeed205 | 26 Aug, 2019, 07:49: PM
Given:
Mass of HCl = 18.25 gm
Molar mass of HCl = 36.5 g/mol
Mass of water = 500 gm
= 0.5 kg
No. of moles of HCl;

Molality is,

Molality of solution is 1 m.
Answered by Varsha | 27 Aug, 2019, 09:56: AM
Application Videos
Concept Videos
JEE main - Chemistry
Asked by ashwinskrishna2006 | 18 Apr, 2024, 05:37: PM
JEE main - Chemistry
Asked by gmafia618 | 04 Apr, 2024, 08:48: PM
JEE main - Chemistry
Asked by jadhavshivtej256 | 27 Feb, 2024, 06:25: PM
JEE main - Chemistry
Asked by pradumankumarsah1 | 30 Jan, 2024, 02:36: PM
JEE main - Chemistry
Asked by srujan11042008 | 06 Nov, 2023, 10:31: AM
JEE main - Chemistry
Asked by vuppulojusaritha | 05 Nov, 2023, 02:22: PM
JEE main - Chemistry
Asked by radheshyambaheti085 | 09 Aug, 2023, 07:10: AM
JEE main - Chemistry
Asked by | 17 Aug, 2022, 08:10: PM
JEE main - Chemistry
Asked by aryankatiyar223 | 10 Aug, 2022, 11:57: PM