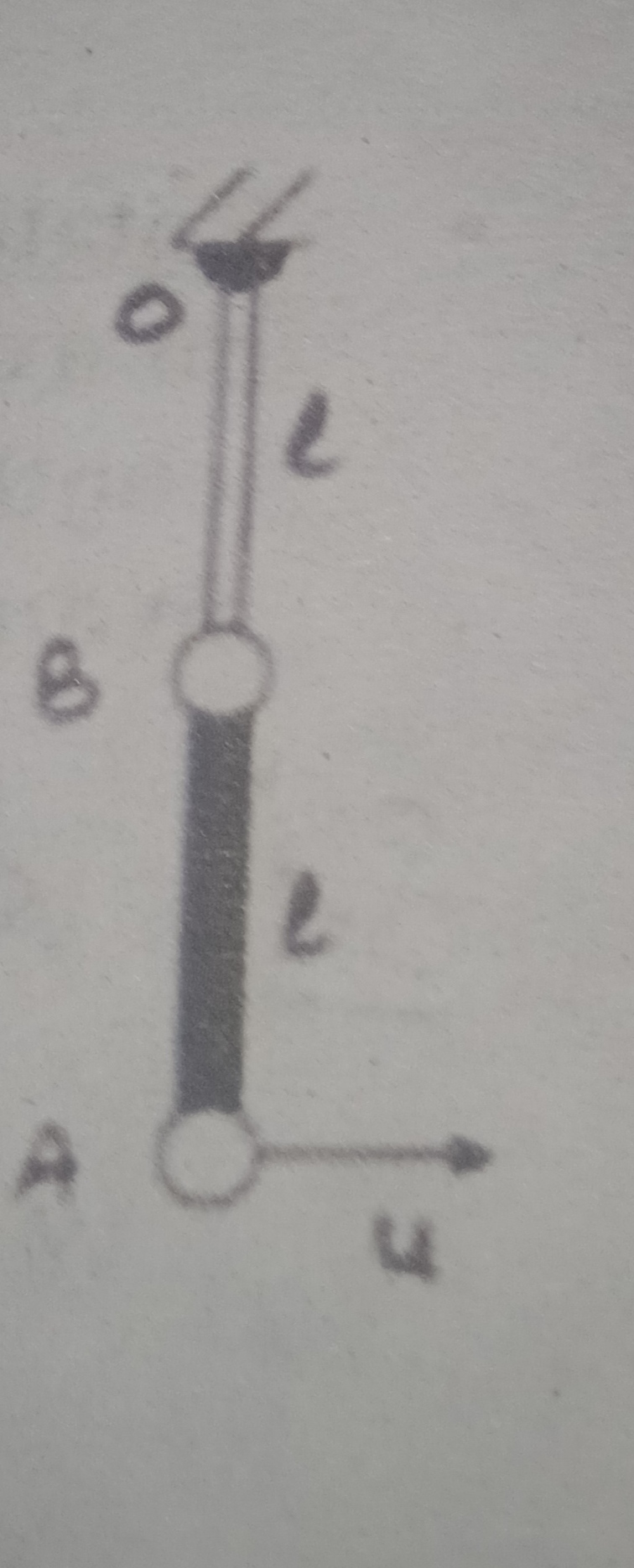CBSE Class 12-science Answered
Surface tension
Asked by | 06 Feb, 2010, 12:16: AM
a) will rise to maximum height.
The water molecules due to force of adhesion clings to the walls of capillary, and since the weight of water in capillary is all that the adhesion can support, the water height in capillary stabilizes.
But since weightless means, no such weight support, then the adhesion will dominate and the water will rise to the maximum height.
Regards,
Team,
TopperLearning.
Answered by | 06 Feb, 2010, 09:50: AM
Concept Videos
CBSE 12-science - Physics
Asked by artabandhusahu85 | 24 Apr, 2024, 12:07: PM
CBSE 12-science - Physics
Asked by niharvijayvargiya5 | 23 Apr, 2024, 06:40: PM
CBSE 12-science - Physics
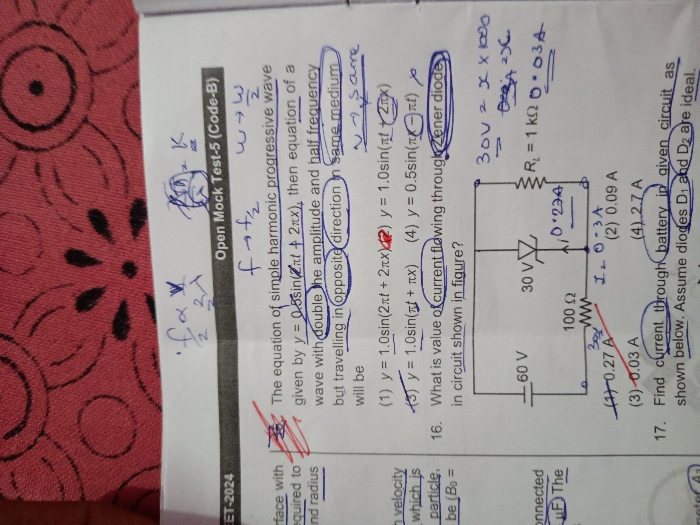
Asked by kulhariabhijeet | 21 Apr, 2024, 02:39: PM
CBSE 12-science - Physics
Asked by mohapatraswetalina88 | 21 Apr, 2024, 12:18: PM
CBSE 12-science - Physics
Asked by aishaisha091098 | 19 Apr, 2024, 04:54: PM
CBSE 12-science - Physics
Asked by dasrituparna1999 | 13 Apr, 2024, 06:56: AM
CBSE 12-science - Physics
Asked by dasrituparna1999 | 12 Apr, 2024, 09:26: PM
CBSE 12-science - Physics
Asked by mishrigupta19319 | 08 Apr, 2024, 06:28: PM
CBSE 12-science - Physics
Asked by madhav9119887644 | 07 Apr, 2024, 08:10: PM