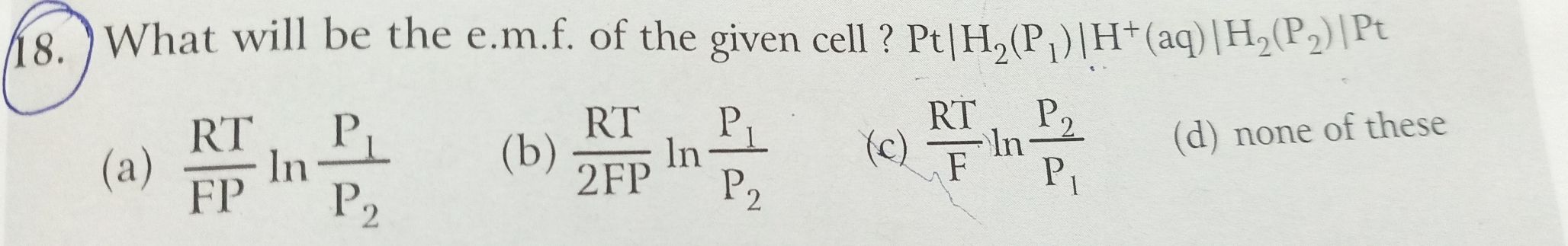JEE Class main Answered
Which of the following will form a cell with highest voltage?
(E0Co2+/Co=−0.28V;E0Ag+/Ag=0.80V)
A.
1M Ag+ , 1M Co2+
B.
2M Ag+ , 2M Co2+
C.
0.1M Ag+ , 2M Co2+
D.
2MA Ag+ , 0.1M Co2+
Asked by s.ojaswini17 | 16 Feb, 2019, 13:02: PM
Option D is correct.
The cell reaction is
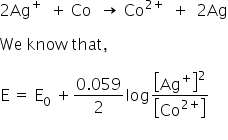
From the equation,
Smaller the concentration of [Co2+] and larger the concentration of [Ag+] will produce maximum e.m.f.
So the correct option is D, 2 M Ag+ and 0.1 M Co2+.
Answered by Varsha | 18 Feb, 2019, 11:41: AM
JEE main - Chemistry
Asked by Balbir | 28 Aug, 2019, 21:05: PM
JEE main - Chemistry
Asked by s.ojaswini17 | 16 Feb, 2019, 13:02: PM
JEE main - Chemistry
Asked by s.ojaswini17 | 15 Feb, 2019, 21:09: PM