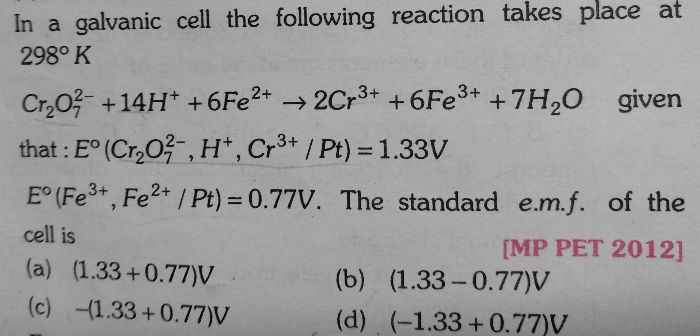NEET Class neet Answered
in fuel cell h2 and o2 rea
Asked by dshreya247 | 03 Feb, 2024, 11:32: AM
Dear Student,
Fuel cells Reactions are as follows:
At anode : 2 H2(g) + 4OH– (aq) → 4H2O(l) + 4e–
At cathode : O2(g) + 2H2O(l) + 4e– → 4 OH– (aq)
Overall reaction: 2 H2(g) + O2(g) → 2H2O(l)
Answered by | 04 Feb, 2024, 18:32: PM
NEET neet - Chemistry
Asked by sr8834055 | 20 Mar, 2024, 14:54: PM
NEET neet - Chemistry
Asked by dshreya247 | 03 Feb, 2024, 11:32: AM
NEET neet - Chemistry
Asked by sujitjana971 | 15 Dec, 2022, 20:09: PM
NEET neet - Chemistry
Asked by zoyakhan98264 | 16 Jul, 2022, 13:59: PM
NEET neet - Chemistry
Asked by gurugubellisaivishal2705 | 30 Jun, 2022, 12:32: PM
NEET neet - Chemistry
Asked by ansh.bharso | 28 Jun, 2022, 15:33: PM
NEET neet - Chemistry
Asked by dev28011997 | 09 Oct, 2021, 14:21: PM
NEET neet - Chemistry
Asked by Prashant DIGHE | 22 Aug, 2020, 21:43: PM
NEET neet - Chemistry
Asked by Prashant DIGHE | 22 Aug, 2020, 21:39: PM