JEE Class main Answered
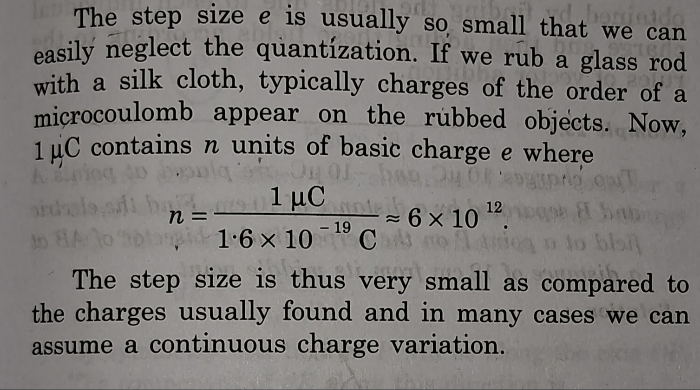
Size of water molecule is 0.3 nm . Hence volume of water molecule is of the order of 10-29 m3 .
If we calculate number n of water molecules in 1 ml of water , we get
n = 10-6 / 10-29 = 1023 molecules .
Moleculer size is so small , if we see 1 ml of water , we see a continuous distribution of molecules throughout the volume of 1 ml.
we do not see discrete molecules even if see through microscope.
But if we see any fine powder , even with naked eye , we see discrete particles of powder.
It is possible to see discrete particles of powder because size of one particle of powder is few orders less than size of powder sample.
For example , if particle size is few microns , then it is possible to see discrete particle in few milli gram sample of powder.
Whereas in few ml of water , we can not see discrete water molecules because size of volume ratio between one molecule and
volume sample is very huge ( order of 1023 ) .
Similarly in the case of charge , fundamental charge which is charge of electron ( 1.6 × 10-19 C ) is so small compare to the charge distribution
like charges developed on glass surface if it rubbed with silk cloth ( order of μC ) .
Hence we do not see discrete charges in the charge distribution but continuous distribution .