CBSE Class 12-science Answered
figure for 13.6 in chapter nuclei
Asked by silladech | 16 Nov, 2018, 11:14: AM
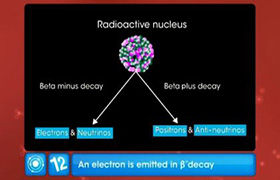
This diagram is showing the decay of an atom.
For gamma decay frequencies u just need to find the energy difference between energy level.
Like for γ1 energy difference is 1.088-0= 1.088
So the energy of photon is 1.088MeV
hf = 1.088Mev here h is Planck constant and f us frequency. This way we can find the frequency for any gamma decay
For beta decay
We just have to find the energy difference
Change in mass = 197.968233 - 197.966760 =0.001473u
1u= 931.5 MeV
Then energy = 931.5×0.001473=1.3720995
Change in energy = kinetic energy of beta particle= 1.088- 1.3720995 =0.2840995 Mev.
Answered by Ankit K | 19 Nov, 2018, 22:16: PM
Concept Videos
CBSE 12-science - Physics
Asked by prithviraj.chopra2011 | 14 Jul, 2024, 23:00: PM
CBSE 12-science - Physics
Asked by arjunsah797 | 16 May, 2022, 14:17: PM
CBSE 12-science - Physics
Asked by merinlijo_20002 | 17 Jun, 2020, 10:05: AM
CBSE 12-science - Physics
Asked by rajubarman | 01 Dec, 2019, 10:03: AM
CBSE 12-science - Physics
Asked by alanpeter9611 | 23 Feb, 2019, 19:47: PM
CBSE 12-science - Physics
Asked by alanpeter9611 | 22 Feb, 2019, 23:24: PM
CBSE 12-science - Physics
Asked by sd2021667 | 04 Dec, 2018, 16:46: PM
CBSE 12-science - Physics
Asked by silladech | 16 Nov, 2018, 11:14: AM
CBSE 12-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 13:23: PM
CBSE 12-science - Physics
Asked by Topperlearning User | 09 Jul, 2014, 16:06: PM






 , find the number of nuclei present after 8 days?
, find the number of nuclei present after 8 days?