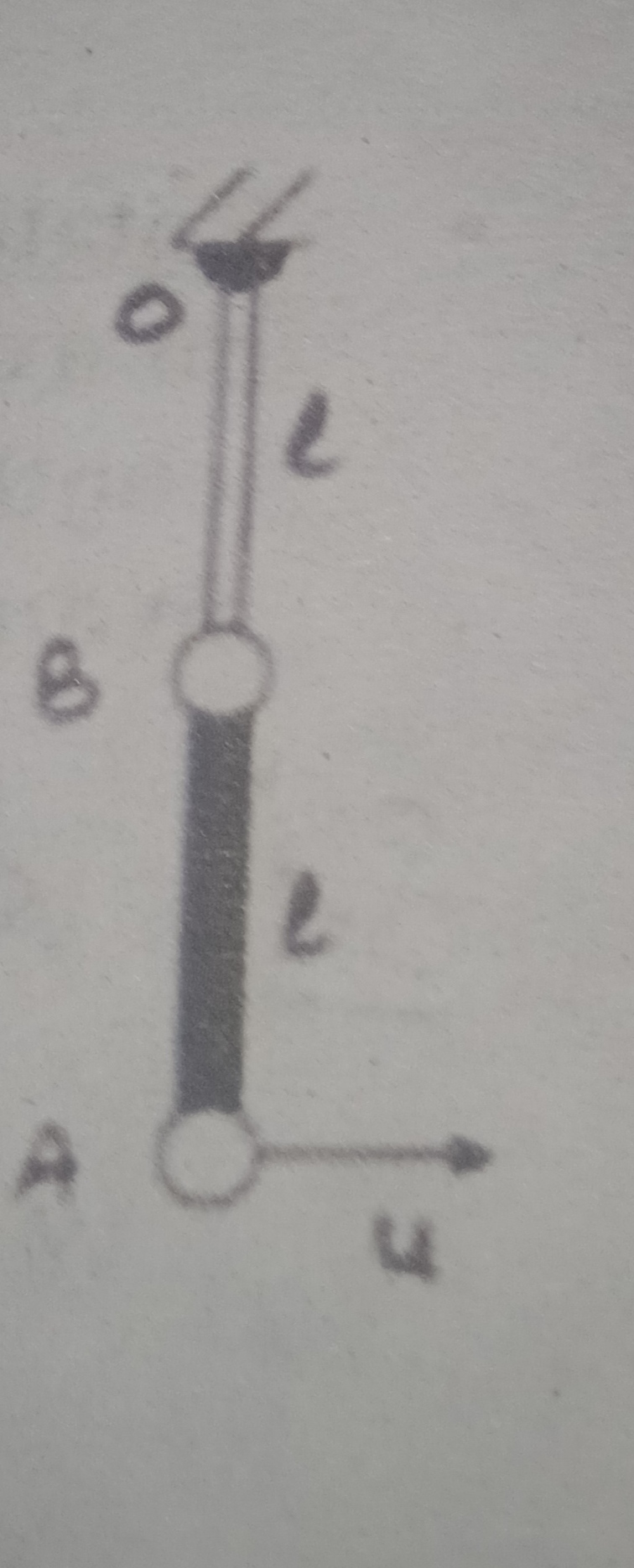CBSE Class 12-science Answered
Describe the experiment to study photoelectric effect and explain the laws of photoelectric effect and the significance of each
Asked by subashchoudhary0099 | 01 Mar, 2021, 11:42: PM
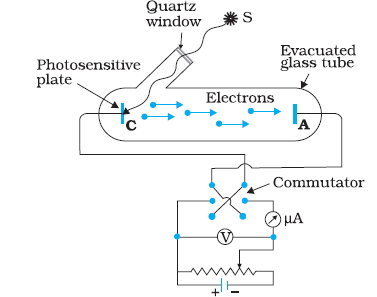
Figure given above depicts a schematic view of the arrangement used for the experimental study of the photoelectric effect.
It consists of an evacuated glass/quartz tube having a photosensitive plate C and another metal plate A.
Monochromatic light from the source S of sufficiently short wavelength passes through the window W and falls on the
photosensitive plate C (emitter). A transparent quartz window is sealed on to the glass tube, which permits ultraviolet radiation
to pass through it and irradiate the photosensitive plate C. The electrons are emitted by the plate C and are collected by
the plate A (collector), by the electric field created by the battery.
The battery maintains the potential difference between the plates C and A, that can be varied.
The polarity of the plates C and A can be reversed by a commutator. Thus, the plate A can be maintained at a desired
positive or negative potential with respect to emitter C. When the collector plate A is positive with respect to the emitter plate C,
positive or negative potential with respect to emitter C. When the collector plate A is positive with respect to the emitter plate C,
the electrons areattracted to it. The emission of electrons causes flow of electric current in the circuit. The potential difference
between the emitter and collector plates is measured by a voltmeter (V) whereas the resulting photo current
flowing in the circuit is measured by a microammeter (μA). The photoelectric current can be increased or decreased by
between the emitter and collector plates is measured by a voltmeter (V) whereas the resulting photo current
flowing in the circuit is measured by a microammeter (μA). The photoelectric current can be increased or decreased by
varying the potential of collector plate A with respect to the emitter plate C. The intensity and frequency of the incident light
can be varied, as can the potential difference V between the emitter C and the collector A.
The experimental arrangement is used to study the variation of photocurrent with
(a) intensity of radiation,
(a) intensity of radiation,
(b) frequency of incident radiation,
(c) the potential difference between the plates A and C, and
(d) the nature of the material of plate C.
Light of different frequencies can be used by putting appropriate coloured filter or coloured glass in the path of light falling
on the emitter C. The intensity of light is varied by changing the distance of the light source from the emitter.
(1) Effect of intensity of light on photocurrent
The collector A is maintained at a positive potential with respect to emitter C so that electrons ejected from C are
attracted towards collector A. Keeping the frequency of the incident radiation and the accelerating potential fixed, the
intensity of light is varied and the resulting photoelectric current is measured each time. It is found that the photocurrent
The collector A is maintained at a positive potential with respect to emitter C so that electrons ejected from C are
attracted towards collector A. Keeping the frequency of the incident radiation and the accelerating potential fixed, the
intensity of light is varied and the resulting photoelectric current is measured each time. It is found that the photocurrent
increases linearly with intensity of incident light . The photocurrent is directly proportional to the number of photoelectrons emitted per
second. This implies that the number of photoelectrons emitted per second is directly proportional to the intensity
of incident radiation.
second. This implies that the number of photoelectrons emitted per second is directly proportional to the intensity
of incident radiation.
(2) Effect of potential on photoelectric current
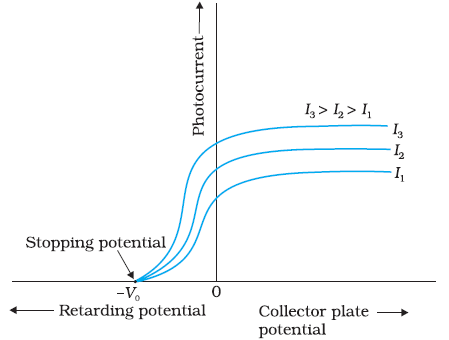
Frequency and intensity of radiation is fixed . When potential difference between emitter and collector is increased ,
It is found that photo electric current gradually increase and reaches saturation value. Since the number of photo electrons
depends upon intensity and enough potential difference is applied to attract all the electrons, photo current reaches saturation .
When collector potential is made negative with respect to emitter, photo current decreases .
After a particular potential that is known as stopping potential , there will not be any photo current . This is because negative
potential retards the electron and electron requires initial kietic energy to reach collector and to give photo current.
Hence negative potential becomes the measure of electron energy . Stopping potential gives the maximum kinetic energy of electron.
It is found in this experiment , Stopping potential is same by varying the intensity of radiation. Stopping potential is independent of
intensity of incident radiation when frequency is fixed . Since stopping potential is measure of maximum kinetic energy of electron ,
when frequency is maintained constant , maximum kinetic energy of electron depends only on material of emitter
but not on intensity of radiation.
( 3 ) Effect of frequency of incident radiation on stopping potential
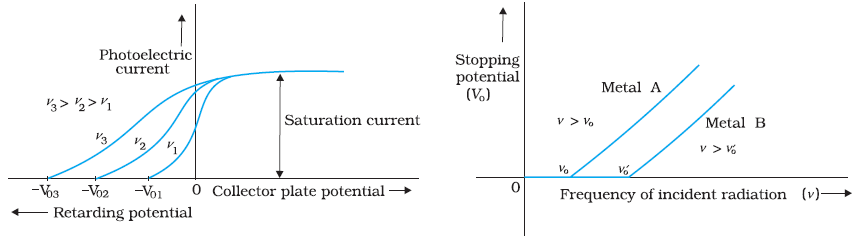
Now if we change the frequency of incident radiation and try to find the stopping potential , it is found that
magnitude of stopping potential increases with increasing frequency of radiation . By decreasing frequency of
radiation stopping potential decreases and reduce to zero at a particular frequency that is known as cutoff frequecny.
Hence from above experiemts we deduce the law of photo electric effect
(i) For a given metal , to get photoelectric current by irradiating the metal with electromagnetic radiation ,
we need minimum frequecy of electromagnetic raditation is required, i.e. knowa as cut off frequency.
Just above cut-off frequency , we get non zero stopping potential. Since stopping potential is measure of kinetic energy
of photo electron and energy of irradiating electromagnetic radiation is proportional to frequncey , we conclude that
minimum energy is required to remove photo electron.
This minimum energy is known as work function of material that is characterstic of material .
Hence we write photo electric equation as, (1/2) m v2 = eVo = ( h  ) - w
) - w
 ) - w
) - wwhere (1/2) m v2 is kinetic energy of electron , e is charge of electron , Vo is stopping potential , h is palancks constant ,
 is frequency of radiation and w is workfunction.
is frequency of radiation and w is workfunction.
Answered by Thiyagarajan K | 02 Mar, 2021, 02:05: PM
Concept Videos
CBSE 12-science - Physics
Asked by panneer1766 | 24 Apr, 2024, 01:52: PM
CBSE 12-science - Physics
Asked by artabandhusahu85 | 24 Apr, 2024, 12:07: PM
CBSE 12-science - Physics
Asked by niharvijayvargiya5 | 23 Apr, 2024, 06:40: PM
CBSE 12-science - Physics
Asked by kulhariabhijeet | 21 Apr, 2024, 02:39: PM
CBSE 12-science - Physics
Asked by mohapatraswetalina88 | 21 Apr, 2024, 12:18: PM
CBSE 12-science - Physics
Asked by aishaisha091098 | 19 Apr, 2024, 04:54: PM
CBSE 12-science - Physics
Asked by dasrituparna1999 | 13 Apr, 2024, 06:56: AM
CBSE 12-science - Physics
Asked by dasrituparna1999 | 12 Apr, 2024, 09:26: PM
CBSE 12-science - Physics
Asked by mishrigupta19319 | 08 Apr, 2024, 06:28: PM