ICSE Class 9 Answered
calculate the percentage of phosphorus in :
(a) Calcium hydrogen phosphate
Ca(H2PO4)2
(a) Calcium hydrogen phosphate
( Ca(
Asked by mtomar7274 | 03 Jun, 2020, 08:26: AM
Calcium hydrogen phosphate Ca(H2PO4)2
Molecular mass of Ca(H2PO4)2 = 234
Atomic weight of P = 31
% of P in Ca(H2PO4)2 = 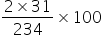
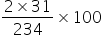
= 26.49 % of P
Answered by Varsha | 03 Jun, 2020, 12:04: PM
Concept Videos
ICSE 9 - Chemistry
Asked by gangolykavita890 | 08 Mar, 2024, 08:32: PM
ICSE 9 - Chemistry
Asked by mangalgourihebballi | 27 Oct, 2023, 12:32: PM
ICSE 9 - Chemistry
Asked by ootysmh1 | 21 Dec, 2022, 09:03: PM
ICSE 9 - Chemistry
Asked by ayantanchoudhuri | 26 Jun, 2022, 06:47: PM
ICSE 9 - Chemistry
Asked by mantashatakey786 | 23 Oct, 2021, 09:53: PM
ICSE 9 - Chemistry
Asked by cdiltaj | 22 Sep, 2021, 08:42: AM
ICSE 9 - Chemistry
Asked by happyddl2012 | 24 Jun, 2021, 10:10: AM
ICSE 9 - Chemistry
Asked by abdulrahim22554466 | 05 Mar, 2021, 01:04: PM
ICSE 9 - Chemistry
Asked by manjudarjee90 | 31 Aug, 2020, 04:05: PM
ICSE 9 - Chemistry
Asked by hemanginivyas92.9spicertl | 14 Aug, 2020, 01:05: PM




