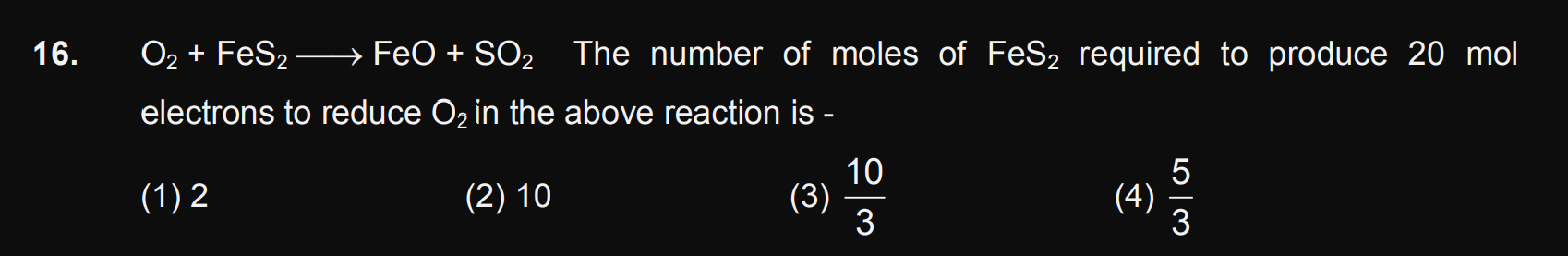JEE Class main Answered
At temp T a co
mpound AB2(g) dissociates according to the reaction
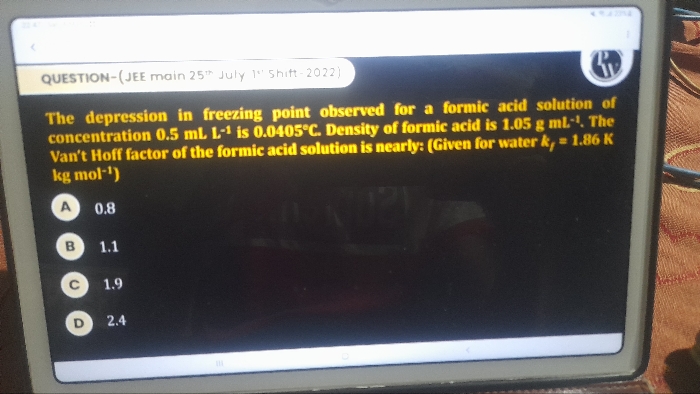
2AB2→2AB+B2 with a degree of dissociation X which is small as compared to unity .the expression for Kpin terms of X and total pressure P is
Asked by ak020820002 | 05 Feb, 2019, 12:48: AM
The compound AB2(g) dissociates according to the reaction,
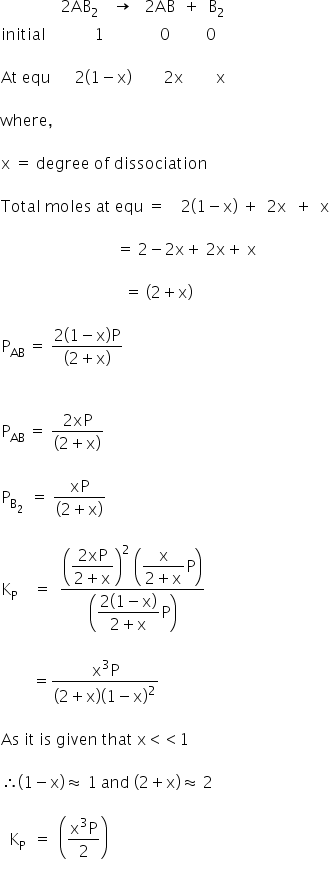
The equilibrium constant Kp = (x3P)/2
Answered by Varsha | 05 Feb, 2019, 11:25: AM
Application Videos
Concept Videos
JEE main - Chemistry
Asked by manishguptaballia.15 | 11 May, 2024, 03:57: PM
JEE main - Chemistry
Asked by srkgb8018 | 10 May, 2024, 07:36: AM
JEE main - Chemistry
Asked by jacksparrow7352133590 | 09 May, 2024, 08:20: PM
JEE main - Chemistry
Asked by purnendurai26 | 02 May, 2024, 06:34: PM
JEE main - Chemistry
Asked by cheekatiyogendra143 | 20 Apr, 2024, 11:16: AM
JEE main - Chemistry
Asked by jwhhebbb | 19 Apr, 2024, 01:21: PM
JEE main - Chemistry
Asked by ashwinskrishna2006 | 18 Apr, 2024, 09:44: PM
JEE main - Chemistry
Asked by ashwinskrishna2006 | 18 Apr, 2024, 05:37: PM