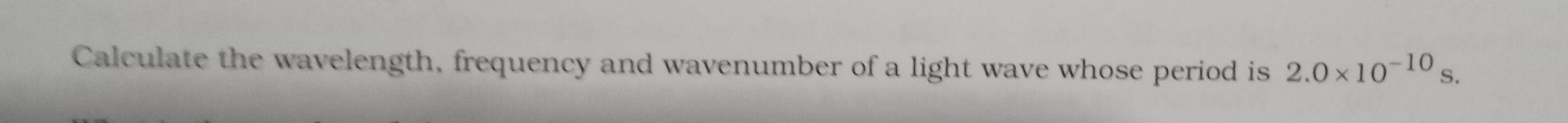JEE Class main Answered
Q. 59
Review Later
If the bond order in Cl O 4 ^ - is x, find the value of 100x.

Asked by cheekatiyogendra143 | 20 Apr, 2024, 11:16: AM
Dear Student,
Electronic configuration of chlorine (atomic number 17) : [Ne]3s23p5
In the first excited state the electronic configuration: 1s22s22p63s23p43d1
In the second excited state the electronic configuration: 1s22s22p63s23p33d2
There are five unpaired electrons in the second excited state of chlorine.
Therefore, the covalency of chlorine in the second excited state will be 5.
Answered by | 22 Apr, 2024, 11:19: AM
JEE main - Chemistry
Asked by radham6375 | 17 May, 2024, 20:13: PM
JEE main - Chemistry
Asked by 9079344910choudhary | 16 May, 2024, 18:52: PM
JEE main - Chemistry
Asked by purnendurai26 | 02 May, 2024, 18:34: PM
JEE main - Chemistry
Asked by cheekatiyogendra143 | 20 Apr, 2024, 11:16: AM
JEE main - Chemistry
Asked by jwhhebbb | 19 Apr, 2024, 13:21: PM
JEE main - Chemistry
Asked by adityadoodi3 | 05 Apr, 2024, 23:27: PM
JEE main - Chemistry
Asked by pratap62437 | 19 Feb, 2024, 12:48: PM
JEE main - Chemistry
Asked by sayushman087 | 01 Feb, 2024, 10:28: AM
JEE main - Chemistry
Asked by marthalamanoharreddy65 | 17 Dec, 2023, 10:26: AM