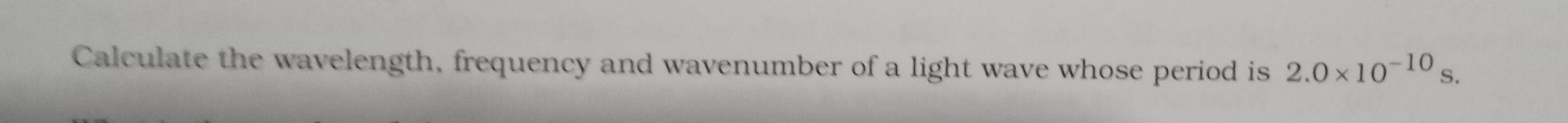JEE Class main Answered
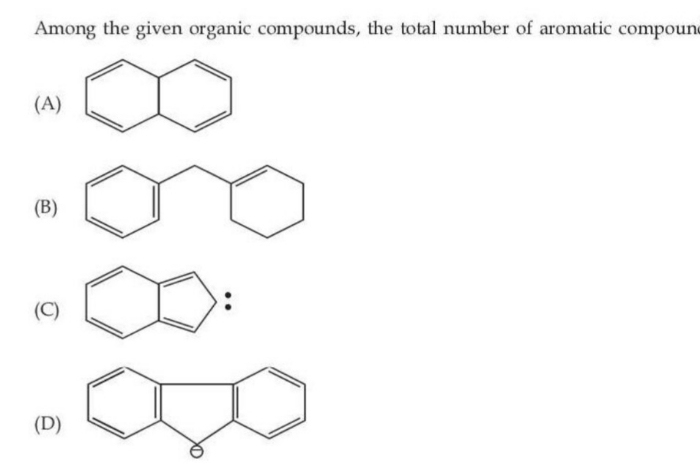
the total no of aromatic compounds
Asked by malayakdha | 07 Feb, 2024, 12:30: PM
Dear Student,
The correct answer option is D which can show aromaticity.
For the cyclic compound to be aromatic, it has to obey 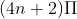 rule.
rule.
Options A and B with only pi binds do not follow this rule.
Also between options C and D, if lone pair electrons mean sp2 hybridized carbon atom being a part of the ring, if not contributing to aromaticity then they should not be considered for 4n+2 rule.
Considering this fact, the option C structure does not show aromaticity.
Answered by | 07 Feb, 2024, 05:25: PM
Application Videos
Concept Videos
JEE main - Chemistry
Asked by cheekatiyogendra143 | 20 Apr, 2024, 11:16: AM
JEE main - Chemistry
Asked by jwhhebbb | 19 Apr, 2024, 01:21: PM
JEE main - Chemistry
Asked by adityadoodi3 | 05 Apr, 2024, 11:27: PM
JEE main - Chemistry
Asked by pratap62437 | 19 Feb, 2024, 12:48: PM
JEE main - Chemistry
Asked by malayakdha | 07 Feb, 2024, 12:30: PM
JEE main - Chemistry
Asked by sayushman087 | 01 Feb, 2024, 10:28: AM
JEE main - Chemistry
Asked by marthalamanoharreddy65 | 17 Dec, 2023, 10:26: AM
JEE main - Chemistry
Asked by aftab01561 | 02 Jul, 2022, 11:35: PM
JEE main - Chemistry
Asked by katakamsettygayathri | 11 Jun, 2022, 09:49: PM
JEE main - Chemistry
Asked by vkanirudh2 | 30 May, 2022, 09:58: PM