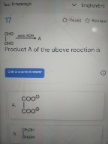
NEET Class neet Answered
Ques: What is the basic principle of conductometric titration?
A.Difference in the relative speed of the ions based on charge
B.Difference in the relative speed of the ions based on charge and mass
C.Partial neutralization reaction
D.Complex formation reactions
Asked by shruti.tripathi2002 | 13 Feb, 2021, 06:33: PM
The principle of the conductometric titration process can be stated as follows – During a titration process, one ion is replaced with another and the difference in the ionic conductivities of these ions directly impacts the overall electrolytic conductivity of the solution.
Answered by Ramandeep | 15 Feb, 2021, 01:45: PM
Application Videos
Concept Videos
NEET neet - Chemistry
Asked by dikshitgod07 | 29 Apr, 2024, 08:38: AM
NEET neet - Chemistry
Asked by biswassayan8464 | 21 Apr, 2024, 11:30: AM
NEET neet - Chemistry
Asked by mahendar160786 | 16 Apr, 2024, 09:23: PM
NEET neet - Chemistry
Asked by raomayankup83 | 15 Apr, 2024, 07:46: PM
NEET neet - Chemistry
Asked by muskannawab11 | 14 Apr, 2024, 03:13: PM
NEET neet - Chemistry
Asked by 8239682116rahul | 10 Apr, 2024, 01:48: PM
NEET neet - Chemistry
Asked by tarasingrathod63 | 07 Apr, 2024, 01:07: PM
NEET neet - Chemistry
Asked by fathimahusna6122 | 05 Apr, 2024, 10:25: AM
NEET neet - Chemistry
Asked by vasantagomasi23 | 05 Apr, 2024, 08:35: AM