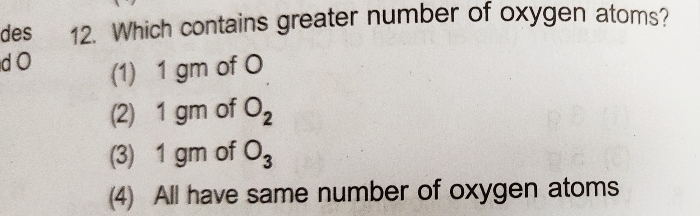NEET Class neet Answered
what is the molarity of solution of 5.85 gram NaCl present in 100 ml solution
Asked by mahendar160786 | 16 Apr, 2024, 21:23: PM
Dear Student,
Molarity = number of moles / volume of solution
M = n / V
Number of moles, n = mass / molar mass
Molar mass of NaCl = 58.5 g/mol
Mass = 5.85 g
n = 5.85 / 58.5 = 0.1 mol
Volume, V = 100 mL = 0.1 L
∴ Molarity = 0.1 / 0.1 = 1 mol/L = 1 M
Answered by | 17 Apr, 2024, 09:59: AM
NEET neet - Chemistry
Asked by kishusambhar | 05 Jun, 2024, 11:31: AM
NEET neet - Chemistry
Asked by priyankasahoo0086 | 22 May, 2024, 06:36: AM
NEET neet - Chemistry
Asked by sahumansi | 12 May, 2024, 09:37: AM
NEET neet - Chemistry
Asked by biswassayan8464 | 21 Apr, 2024, 11:30: AM
NEET neet - Chemistry
Asked by mahendar160786 | 16 Apr, 2024, 21:23: PM
NEET neet - Chemistry
Asked by muskannawab11 | 14 Apr, 2024, 15:13: PM
NEET neet - Chemistry
Asked by tarasingrathod63 | 07 Apr, 2024, 13:07: PM
NEET neet - Chemistry
Asked by fathimahusna6122 | 05 Apr, 2024, 10:25: AM
NEET neet - Chemistry
Asked by vaka.aruna1979 | 23 Mar, 2024, 04:18: AM
NEET neet - Chemistry
Asked by fathimahusna23042004 | 03 Mar, 2024, 08:56: AM