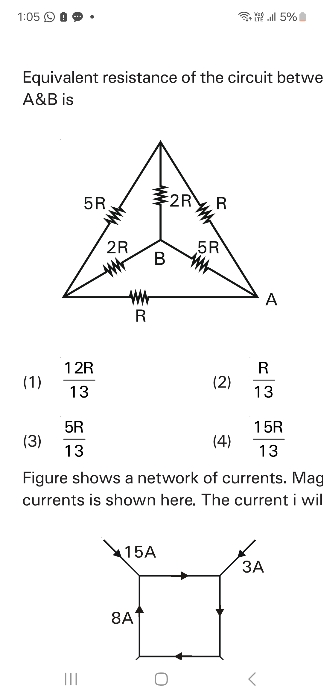
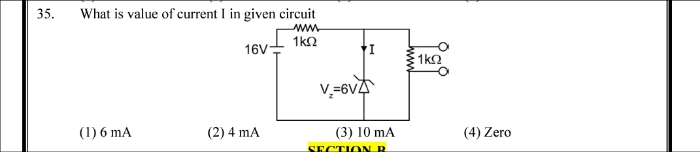
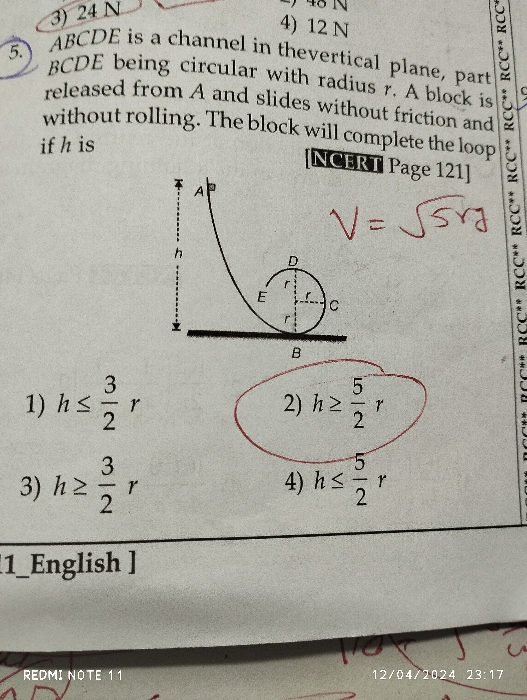NEET Class neet Answered
please answer with expalnation
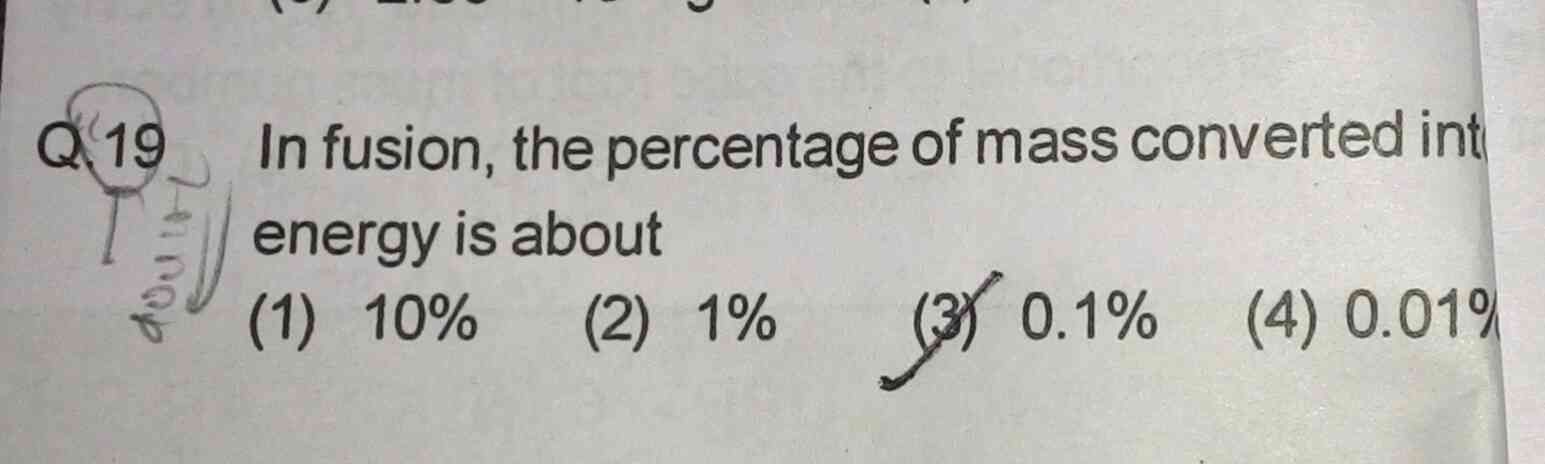
Asked by Prashant DIGHE | 16 Jan, 2020, 10:00: PM
There are many number of fusion reactions.
Let us take the most favourable Deuterium-Tritium fusion reaction
2H1 + 3H1 → 4He2 + 1n0 + 17.59 MeV
Total mass of reactants = mass of deuterium ( 2.014 amu ) + mass of tritium ( 3.016 amu ) = 5.03 amu
Total mass of products = mass of Helium ( 4.0015 amu ) + mass of neutron ( 1.0073 amu ) = 5.0088amu
Mass difference that appears in the form of energy = ( 5.030 + 5.0088 ) = 0.0212 amu
Hence % of mass converted into energy = (.0212 / 5.03) × 100 = 0.42 %
Answered by Thiyagarajan K | 17 Jan, 2020, 08:27: AM
Application Videos
NEET neet - Physics
Asked by ramanjaneyuluoguru | 25 Apr, 2024, 04:18: PM
NEET neet - Physics
Asked by shatakshibhatt9 | 20 Apr, 2024, 07:52: PM
NEET neet - Physics
Asked by praveenpriya000079 | 18 Apr, 2024, 07:24: AM
NEET neet - Physics
Asked by gouranshi84 | 17 Apr, 2024, 05:23: PM
NEET neet - Physics
Asked by sojusvi | 17 Apr, 2024, 01:12: PM