NEET Class neet Answered
please answer this

Asked by Prashant DIGHE | 16 Feb, 2020, 10:09: PM
option (1) is coorect.
The reaction is:
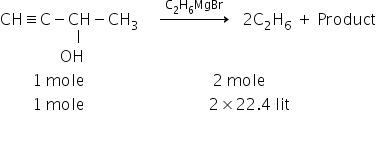
Given:
Mass of but-3 -yn-2-ol = 0.21 gm
Molar mass of but-3 -yn-2-ol = 70
No. of moles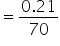
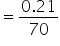
= 0.003 mol
So 0.003 mole of but-3 -yn-2-ol will give
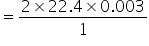
= 0.1344 lit
= 134.4 ml
The volume of gas (C2H6) evolved is 134.4 ml
Answered by Varsha | 17 Feb, 2020, 12:17: PM
NEET neet - Chemistry
Asked by biswassayan8464 | 21 Apr, 2024, 11:30: AM
NEET neet - Chemistry
Asked by mahendar160786 | 16 Apr, 2024, 09:23: PM
NEET neet - Chemistry
Asked by muskannawab11 | 14 Apr, 2024, 03:13: PM
NEET neet - Chemistry
Asked by tarasingrathod63 | 07 Apr, 2024, 01:07: PM
NEET neet - Chemistry
Asked by fathimahusna6122 | 05 Apr, 2024, 10:25: AM
NEET neet - Chemistry
Asked by vaka.aruna1979 | 23 Mar, 2024, 04:18: AM
NEET neet - Chemistry
Asked by fathimahusna23042004 | 03 Mar, 2024, 08:56: AM
NEET neet - Chemistry
Asked by drkeshavkhandagle | 18 Jan, 2024, 08:10: PM
NEET neet - Chemistry
Asked by yogitakumawat | 21 Dec, 2023, 10:31: PM






