NEET Class neet Answered
please answer this

Asked by Prashant DIGHE | 03 Mar, 2020, 10:09: PM
Option (1) is correct.
Given:
Volume of container = 20 L
Temperature, t = 400 K
Pressure = 0.4 atm
The given reaction is,
SrCO3(s) 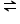 SrO(s) + CO2(g)
SrO(s) + CO2(g)
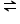 SrO(s) + CO2(g)
SrO(s) + CO2(g) kp = 1.6 atm
PCO2 = 1.6 atm = Maximum pressure of CO2
Volume of conatainer at this stage is;
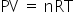
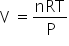 .....(1)
.....(1)n = constant, as reaction was not at equilibrium.
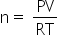
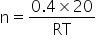 ....(2)
....(2)From eq(1) and (2)
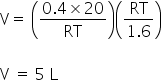
The maximum volume of the container at maximum pressure is 5 L.
Answered by Varsha | 04 Mar, 2020, 01:18: PM
NEET neet - Chemistry
Asked by raomayankup83 | 15 Apr, 2024, 07:46: PM
NEET neet - Chemistry
Asked by gopikakannan27 | 13 Jan, 2022, 05:39: AM
NEET neet - Chemistry
Asked by jhajuhi19 | 19 Aug, 2021, 08:38: PM
NEET neet - Chemistry
Asked by kowsalyamouli | 06 Oct, 2020, 04:08: PM
NEET neet - Chemistry
Asked by nssharma001969 | 03 Jun, 2020, 01:54: AM
NEET neet - Chemistry
Asked by prakriti12oct | 25 May, 2020, 12:15: AM
NEET neet - Chemistry
Asked by jhajuhi19 | 10 May, 2020, 08:24: AM
NEET neet - Chemistry
Asked by anushka.kulkarni02022002 | 19 Mar, 2020, 09:31: AM
NEET neet - Chemistry
Asked by Prashant DIGHE | 03 Mar, 2020, 10:09: PM
NEET neet - Chemistry
Asked by Prashant DIGHE | 03 Mar, 2020, 10:06: PM