NEET Class neet Answered
how catalyst does not catalyse nonspontaneous reaction ?
Asked by baloutpreety | 25 Dec, 2018, 11:26: AM
Non Spontaneous reaction requires the force in the form of pressure, heat etc to proceed so they are endothermic in nature. Catalyst to function needs to be activated and in case of non spontaneous reaction catalyst do not get the environment to activate its surface so is non functional.
Answered by Sumit Chakrapani | 26 Dec, 2018, 02:50: AM
Application Videos
Concept Videos
NEET neet - Chemistry
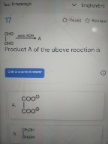
Asked by biswassayan8464 | 21 Apr, 2024, 11:30: AM
NEET neet - Chemistry
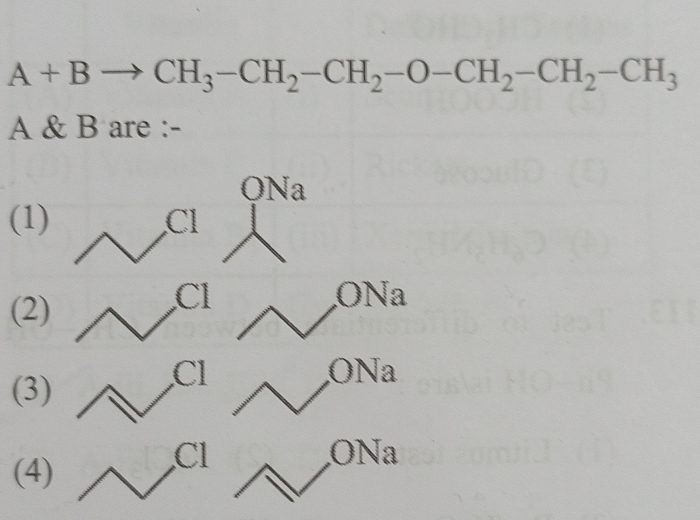
Asked by mahendar160786 | 16 Apr, 2024, 09:23: PM
NEET neet - Chemistry
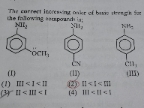
Asked by raomayankup83 | 15 Apr, 2024, 07:46: PM
NEET neet - Chemistry
Asked by muskannawab11 | 14 Apr, 2024, 03:13: PM
NEET neet - Chemistry
Asked by 8239682116rahul | 10 Apr, 2024, 01:48: PM
NEET neet - Chemistry
Asked by tarasingrathod63 | 07 Apr, 2024, 01:07: PM
NEET neet - Chemistry
Asked by fathimahusna6122 | 05 Apr, 2024, 10:25: AM
NEET neet - Chemistry
Asked by vasantagomasi23 | 05 Apr, 2024, 08:35: AM
NEET neet - Chemistry
Asked by ankuruthanuriya | 03 Apr, 2024, 10:56: PM