NEET Class neet Answered
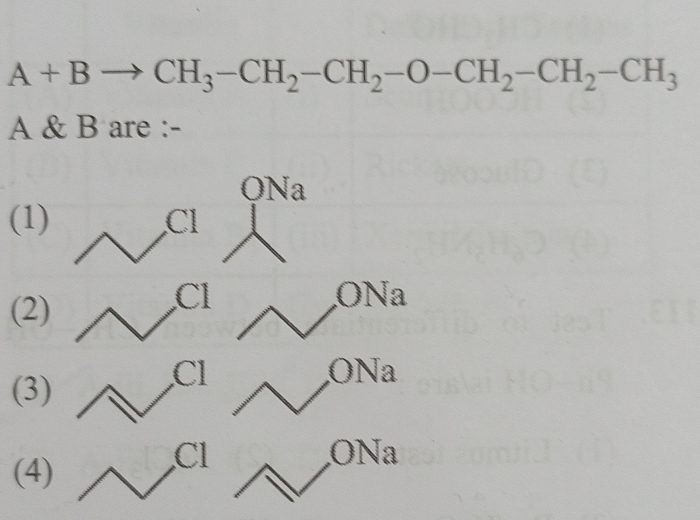
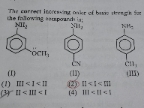
explain

Asked by myindiaisbad | 14 Feb, 2023, 02:12: AM
Dear Student,
Mass of solute = 18 g
Molar mass of benzene = 78.11 g/mol
P = 80% P0 = 0.8 P0
Relative lowering of vapour pressure = (P0 – P) / P0 = XB = Mole fraction of solute
(P0 – 0.8 P0) / P0 = nB / (nA + nB)
0.2 = nB / (nA + nB)
But number of solute particles is very less compare to solvent particles, hence
0.2 = nB / nA
Mole of solvent = 117 / 78.11 ≈ 1.50
Let molar mass of solute be ‘Y’
0.2 = (18 / Y) / 1.50
0.3 = 18/Y
Y = 60 g
Molar mass of solute is 60 g.
In the given answer options, the correct answer is missing.
Answered by | 14 Feb, 2023, 02:26: PM
Application Videos
Concept Videos
NEET neet - Chemistry
Asked by biswassayan8464 | 21 Apr, 2024, 11:30: AM
NEET neet - Chemistry
Asked by mahendar160786 | 16 Apr, 2024, 09:23: PM
NEET neet - Chemistry
Asked by raomayankup83 | 15 Apr, 2024, 07:46: PM
NEET neet - Chemistry
Asked by muskannawab11 | 14 Apr, 2024, 03:13: PM
NEET neet - Chemistry
Asked by 8239682116rahul | 10 Apr, 2024, 01:48: PM
NEET neet - Chemistry
Asked by tarasingrathod63 | 07 Apr, 2024, 01:07: PM
NEET neet - Chemistry
Asked by fathimahusna6122 | 05 Apr, 2024, 10:25: AM
NEET neet - Chemistry
Asked by vasantagomasi23 | 05 Apr, 2024, 08:35: AM
NEET neet - Chemistry
Asked by ankuruthanuriya | 03 Apr, 2024, 10:56: PM