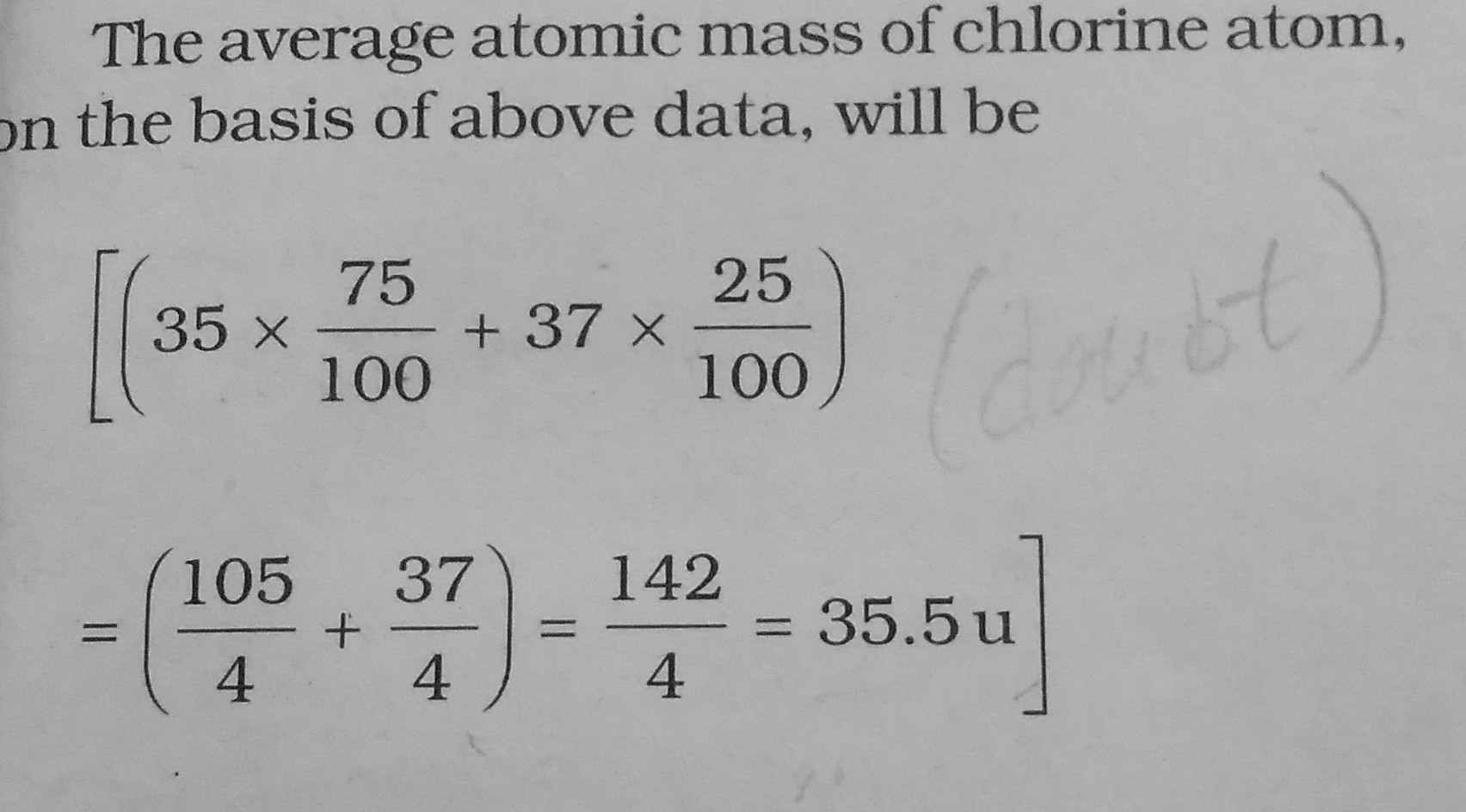CBSE Class 9 Answered
drawback of bohr's atomic model
Asked by shivalaxmi0205 | 08 Mar, 2023, 07:47: PM
Drawbacks in Bohr's atom model is
(1) It does not explain zeema effect and stark effect of atomic spectra
( zeeman effect is splitting of spectral lines of atoms in magnetic field . Stark effect is splitting of spectral lines in electric field )
(2) It could not explain the spectra obtained from larger atoms.
(3) Bohr’s theory is applicable to hydrogen-like species containing one electron only e.g. Singly ionised helium atom
Answered by Thiyagarajan K | 09 Mar, 2023, 09:34: AM
CBSE 9 - Science
Asked by forever.neeta | 19 Jan, 2024, 06:59: PM
CBSE 9 - Science
Asked by siddharthsingha2009 | 07 Jan, 2024, 05:35: AM
CBSE 9 - Science
Asked by sagrawal0081 | 17 Aug, 2023, 05:38: PM
CBSE 9 - Science
Asked by shivalaxmi0205 | 08 Mar, 2023, 07:47: PM
CBSE 9 - Science
Asked by sreesaimadhurakn | 23 Jul, 2022, 06:21: PM
CBSE 9 - Science
Asked by mallharkapse41.9 | 15 Jul, 2022, 08:07: PM
CBSE 9 - Science
Asked by killadalakshmi10 | 20 Jun, 2022, 10:57: AM
CBSE 9 - Science
Asked by seemadevesh26 | 13 Mar, 2022, 03:20: PM
CBSE 9 - Science
Asked by lkaausik | 09 Jan, 2022, 08:28: AM
CBSE 9 - Science
Asked by PURAVVEKARIYA.soc | 01 Sep, 2021, 04:46: PM