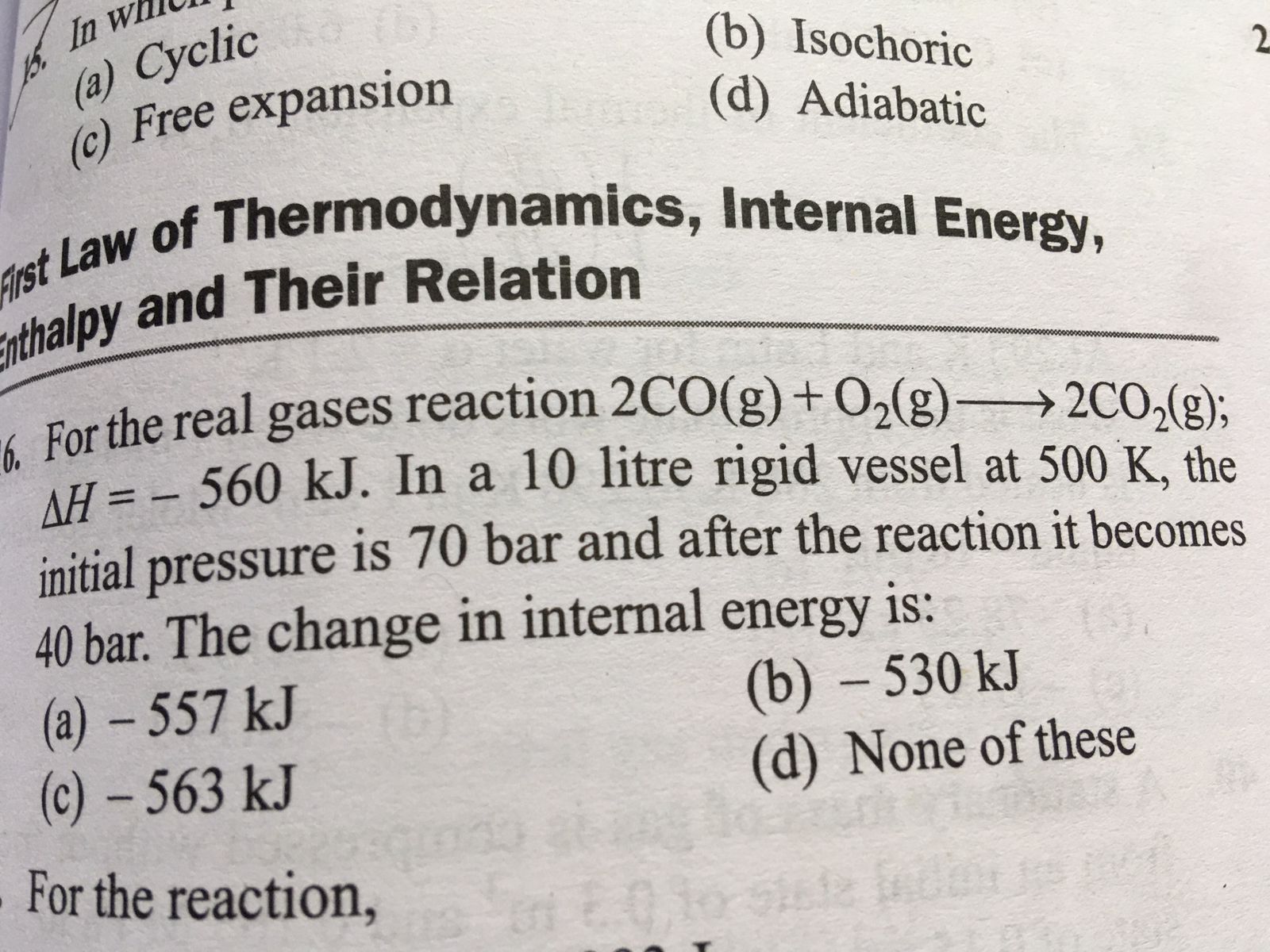NEET Class neet Answered
under isothermal conditions a gas at 300k expand form 0.1l to 0.25l against constant external pressure of 2 bar the work done by the gas is
Asked by deeptideepusingh987 | 23 Oct, 2023, 01:57: PM
1 L-Bar = 100 J
The temperature of the gas, T= 300 K
Initial volume, V1= 0.1 L
Final volume, V2 = 0.25 L
External pressure, P = 2 bar
Work done by the gas, W = ?
A gas expanding against a constant external pressure is an irreversible process.
The work is done in an irreversible process.
W = -PΔV
W = - P (V2 - V1)
Here,
W is the work done.
P is the pressure.
V is the volume.
The negative (-) sign indicates the system loses energy.
By substituting the given values in the above formula, we get
W = -2 Bar (0.25 L – 0.1 L)
= -2 ⨯ 0.15 L Bar
= -0.30 L Bar (1 L Bar = 100 J)
= - 0.30 ⨯ 100 J
= -30 J
Answered by | 24 Oct, 2023, 12:38: PM
NEET neet - Chemistry
Asked by vasantagomasi23 | 05 Apr, 2024, 08:35: AM
NEET neet - Chemistry
Asked by shaiksufiyan.klr | 13 Jan, 2024, 11:49: AM
NEET neet - Chemistry
Asked by mistymouni2507 | 30 Oct, 2023, 10:27: AM
NEET neet - Chemistry
Asked by deeptideepusingh987 | 23 Oct, 2023, 01:57: PM
NEET neet - Chemistry
Asked by priscillaabram | 22 Mar, 2022, 03:59: PM
NEET neet - Chemistry
Asked by meenatoofansingh3 | 03 May, 2021, 04:04: PM
NEET neet - Chemistry
Asked by Prashant DIGHE | 28 Feb, 2020, 10:00: PM
NEET neet - Chemistry
Asked by shwetarajan25 | 05 Dec, 2019, 06:38: AM
NEET neet - Chemistry
Asked by prakriti12oct | 29 Oct, 2019, 12:54: AM
NEET neet - Chemistry
Asked by deepakudgiri29 | 04 Jan, 2019, 05:14: PM