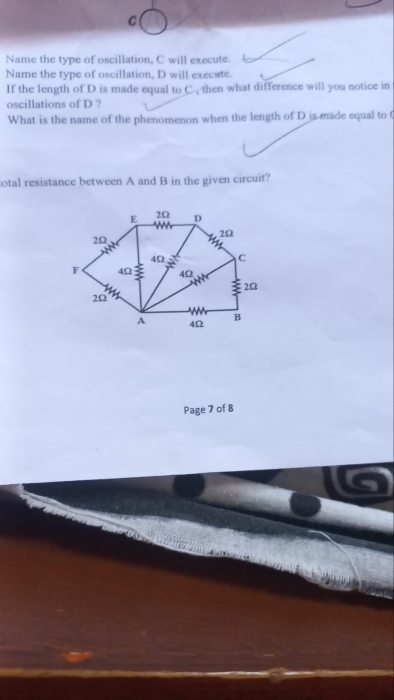
ICSE Class 10 Answered
three electron in p sub level must have the quantum number
Asked by ruchisharmatbn | 24 Feb, 2024, 09:07: PM
p sub level has azimuthal quantum number ( orbital angular momentum ) 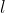 = 1
= 1
Allowed magnetic quantum numbers for sub level that has azimuthal quantum number 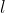 is integr values from -
is integr values from - 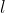 to
to 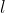
Hence magntic quantum numbers for p sub level are -1 , 0, 1
Hence three electrons in p sub level has magnetic quantum numbers -1, 0, 1
Answered by Thiyagarajan K | 24 Feb, 2024, 09:59: PM
ICSE 10 - Science
Asked by Sammusamreen25 | 06 Mar, 2024, 08:49: PM
ICSE 10 - Science
Asked by ruchisharmatbn | 24 Feb, 2024, 09:07: PM
ICSE 10 - Science
Asked by praveengs1974 | 15 Feb, 2024, 02:11: PM
ICSE 10 - Science
Asked by farzana999 | 10 Feb, 2024, 09:39: PM
ICSE 10 - Science
Asked by saswatamukherjee7c28 | 22 Jan, 2024, 08:48: PM
ICSE 10 - Science
Asked by raysonam111 | 18 Jan, 2024, 07:32: AM
ICSE 10 - Science
Asked by ramgopalsjce | 18 Mar, 2023, 05:54: PM
ICSE 10 - Science
Asked by ramgopalsjce | 18 Mar, 2023, 05:52: PM
ICSE 10 - Science
Asked by aasthamahajan8a3hhps14 | 01 Jan, 2023, 11:40: PM
ICSE 10 - Science
Asked by husain68644 | 29 Dec, 2022, 09:53: PM