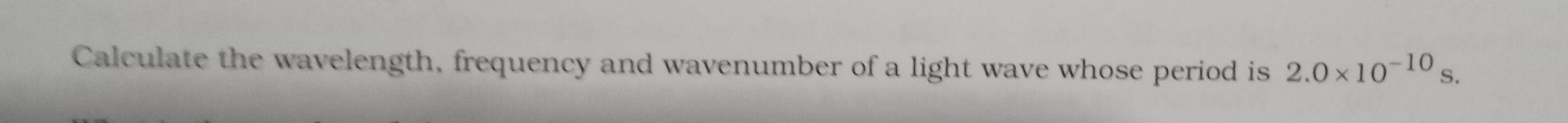JEE Class main Answered
The ionization enery of HE+ is 19.6*10-18 J/ion.Calculate the enery of the first stationary state of Li2+.
Asked by mahadev2471 | 08 Jun, 2019, 11:51: AM
Given:
Ionisation energy of He+ = 19.6 × 10−18 J/ion
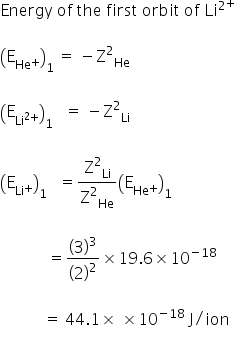
The enery of the first stationary state of Li2+ is 44.1× 10-18 J/ion
Answered by Varsha | 08 Jun, 2019, 22:12: PM
JEE main - Chemistry
Asked by radham6375 | 17 May, 2024, 20:13: PM
JEE main - Chemistry
Asked by 9079344910choudhary | 16 May, 2024, 18:52: PM
JEE main - Chemistry
Asked by purnendurai26 | 02 May, 2024, 18:34: PM
JEE main - Chemistry
Asked by cheekatiyogendra143 | 20 Apr, 2024, 11:16: AM
JEE main - Chemistry
Asked by jwhhebbb | 19 Apr, 2024, 13:21: PM
JEE main - Chemistry
Asked by adityadoodi3 | 05 Apr, 2024, 23:27: PM
JEE main - Chemistry
Asked by pratap62437 | 19 Feb, 2024, 12:48: PM
JEE main - Chemistry
Asked by sayushman087 | 01 Feb, 2024, 10:28: AM
JEE main - Chemistry
Asked by marthalamanoharreddy65 | 17 Dec, 2023, 10:26: AM