CBSE Class 10 Answered
Solutions of barium chloride and sodium sulphate in water react to give insoluble barium sulphate and the solution of sodium chloride
Asked by dheerajvp8055 | 05 May, 2020, 10:57: AM
When an aqueous solution of sodium sulphate reacts with an aqueous solution of barium chloride, insoluble barium sulphate along with solution of sodium chloride is formed. If the reactants are in solid state, then reaction will not take place between sodium sulphate and barium chloride.
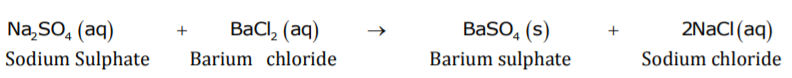
Reaction between aqueous solution of sodium sulphate and aqueous solution of barium chloride is a double displacement reaction.
Answered by Ramandeep | 05 May, 2020, 21:56: PM
Application Videos
Concept Videos
CBSE 10 - Chemistry
Asked by sagarmishra | 04 Mar, 2024, 09:50: AM
CBSE 10 - Chemistry
Asked by susrisangita792 | 17 Nov, 2023, 20:27: PM
CBSE 10 - Chemistry
Asked by pranavvarshansk_9a | 21 May, 2021, 12:06: PM
CBSE 10 - Chemistry
Asked by Visheshsinghvi | 05 May, 2020, 17:54: PM
CBSE 10 - Chemistry
Asked by dheerajvp8055 | 05 May, 2020, 10:57: AM
CBSE 10 - Chemistry
Asked by Visheshsinghvi | 04 May, 2020, 18:43: PM
CBSE 10 - Chemistry
Asked by jarugulaa | 28 Apr, 2020, 10:14: AM
CBSE 10 - Chemistry
Asked by rk92345383 | 19 Apr, 2020, 10:06: AM
CBSE 10 - Chemistry
Asked by s6vsthigade1772004 | 18 Feb, 2020, 21:02: PM
CBSE 10 - Chemistry
Asked by s6vsthigade1772004 | 18 Feb, 2020, 21:00: PM











