CBSE Class 10 Answered
please tell about
1.diamond
2.graphite
3.fullerenes
Asked by anshuman shukla | 08 Nov, 2010, 12:00: AM
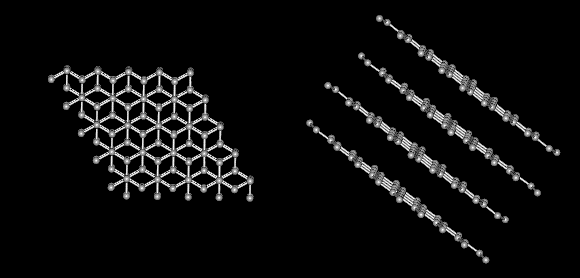
Graphite is very soft and slippery. Diamond is the hardest substance known to man. If both are made only of carbon what gives them different properties? The answer lies in the way the carbon atoms form bonds with each other.
There are strong covalent bonds between carbon atoms in each layer. But, only weak forces exist between layers. This allows layers of carbon to slide overeach other in graphite.
On the other hand, in diamond each carbon atom is the same distance to each of its neighboring carbon atoms. In this rigid network atoms cannot move. This explains why diamonds are so hard and have such a high melting point.
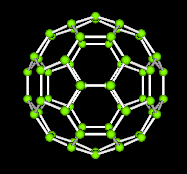
A third class of carbon compounds has recently been discovered. They are called fullerenes. The figure shown is one form composed of 60 carbons. Notice the geometric patterns of pentagons and hexagons that form the familiar icosohedron.
Answered by | 09 Nov, 2010, 05:20: PM
Application Videos
Concept Videos
CBSE 10 - Chemistry
Asked by parthmarch1 | 14 Dec, 2023, 08:27: PM
CBSE 10 - Chemistry
Asked by reetritu34 | 14 Dec, 2023, 07:54: AM
CBSE 10 - Chemistry
Asked by agarwalkrishnam98 | 01 Oct, 2023, 08:28: AM
CBSE 10 - Chemistry
Asked by asra964072 | 18 May, 2022, 10:03: PM
CBSE 10 - Chemistry
Asked by jainnikhil668 | 05 May, 2022, 02:00: PM
CBSE 10 - Chemistry
Asked by gsvjairam | 17 Apr, 2022, 11:32: AM
CBSE 10 - Chemistry
Asked by shubham.sharma80634 | 10 Feb, 2022, 08:43: PM
CBSE 10 - Chemistry
Asked by Trisha Gupta | 25 Jan, 2022, 03:02: PM
CBSE 10 - Chemistry
Asked by Trisha Gupta | 25 Jan, 2022, 03:01: PM
CBSE 10 - Chemistry
Asked by sivaramaraju1000 | 21 Jan, 2022, 09:05: AM