CBSE Class 11-science Answered
if n,l,m are replaced by s,p,q and logic remains the same where s could be 0,2,3,4,...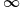 .
.
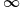 .
.p is defined as 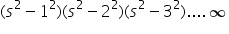 which can also have a value of 0 but no -ive value. q is defined from +p to -p where 0 is not considered in case of fractional value and 1 orbital has 4 electrons . find out:-
which can also have a value of 0 but no -ive value. q is defined from +p to -p where 0 is not considered in case of fractional value and 1 orbital has 4 electrons . find out:-
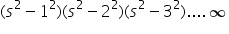 which can also have a value of 0 but no -ive value. q is defined from +p to -p where 0 is not considered in case of fractional value and 1 orbital has 4 electrons . find out:-
which can also have a value of 0 but no -ive value. q is defined from +p to -p where 0 is not considered in case of fractional value and 1 orbital has 4 electrons . find out:-1). Electronic configuration for an atom with an atomic no. of 24 would be.
Asked by Kristin Thapliyal | 11 Aug, 2014, 07:29: PM
Dear nautiyaldivya04@gmail.com
Thanks for asking us a question in Ask the Expert section of TopperLearning.com.
Regards
Topperlearning Team.
Answered by Arvind Diwale | 12 Aug, 2014, 01:50: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by defence | 15 Aug, 2019, 05:24: PM
CBSE 11-science - Chemistry
Asked by drbharatkaila | 25 Nov, 2018, 06:17: PM
CBSE 11-science - Chemistry
Asked by pa1980144 | 14 Oct, 2018, 03:27: PM
CBSE 11-science - Chemistry
Asked by arunavamitra50 | 10 Jun, 2018, 07:17: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 27 Jun, 2016, 12:59: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 10 Sep, 2014, 12:07: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 27 Jun, 2016, 12:59: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 10 Sep, 2014, 12:06: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM




