ICSE Class 10 Answered
Combination Reaction
When two or more substances combine to form a single product, the reaction is known as a combination reaction.
For example:
In the laboratory, iron sulphide is prepared by mixing iron and sulphur.
Fe(s) + S(s) → FeS(s)
- Endothermic Reaction: The reactions accompanied by the absorption of heat are called endothermic reactions.
- Exothermic Reaction: The reactions accompanied by the evolution of heat are called endothermic reactions.
Decomposition Reaction
A chemical reaction in which a single compound splits into two or more simple substances is called a decomposition reaction.
For example:
When mercuric oxide is heated in a crucible, the orange-red powder begins to darken and a silver mirror begins to deposit on the cooler parts of the crucible.
2HgO(s) → 2Hg(s) + O2 ↑
Mercuric oxide Mercury Oxygen
- Thermal Decomposition Reaction: The decomposition reactions carried out by heating are known as thermal decomposition reactions.
- Photochemical reaction: The chemical reactions which proceed with the absorption of light energy are called photochemical reactions.
Displacement Reaction
Reactions in which the more reactive element displaces the less reactive element from its compound are called displacement reactions.
For example:
Zinc displaces copper in copper sulphate to form zinc sulphate.
Zn(s) + CuSO4 (aq) → ZnSO4 (aq) + Cu(s)
Zinc Copper sulphate Zinc sulphate Copper
Double Displacement Reaction
Reactions in which ions of the reactants exchange places to form two new compounds, are called double displacement reactions.
For example:
Sodium hydroxide reacts with hydrochloric acid to form sodium chloride and water.
NaOH(aq) + HCl(aq) → NaCl(aq) + H2O(l)
Oxidation Reactions
Reactions which involve the addition of oxygen or the removal of hydrogen are called oxidation reactions.
For example:
C(s) + 2H2 (g) → CH4 (g)
Reduction Reactions
Chemical reactions in which the reactants gain hydrogen are reduction reactions.
For example:
Fe2O3 + 3CO → 2Fe + 3CO2↑
Ferric oxide Carbon monoxide Iron Carbon dioxide
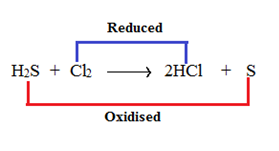
Redox Reaction
The chemical reaction in which oxidation and reduction takes place simultaneously is known as a redox reaction.