ICSE Class 9 Answered
Calculate the temperature to which gas must be heated so that the volume triples without any change in pressure the gas is originally at 57 degree Celsius and having a volume 1 150 cc
Asked by Syedhussain97047 | 14 Feb, 2019, 10:16: PM
From Charle's law we know 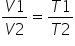
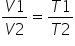
Here V1 = 1.150Ltrs ; V2 = 3 X V1 and T1 = 570C = 330K
Putting the values to above equation we get ,
T2 = 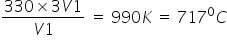
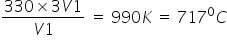
Answered by Sumit Chakrapani | 15 Feb, 2019, 01:53: AM
Application Videos
Concept Videos
ICSE 9 - Chemistry
Asked by sweetsoma1 | 03 May, 2024, 07:29: PM
ICSE 9 - Chemistry
Asked by opkaler802 | 28 Apr, 2024, 07:19: PM
ICSE 9 - Chemistry
Asked by adichikusharma24 | 12 Apr, 2024, 02:36: PM
ICSE 9 - Chemistry
Asked by gangolykavita890 | 08 Mar, 2024, 08:32: PM
ICSE 9 - Chemistry
Asked by shawrina999 | 28 Jan, 2024, 09:57: AM
ICSE 9 - Chemistry
Asked by tripathyshreyam | 29 Dec, 2023, 05:06: PM
ICSE 9 - Chemistry
Asked by aamrinkhanam422 | 24 Dec, 2023, 11:22: AM
ICSE 9 - Chemistry
Asked by zairafathma933 | 22 Dec, 2023, 11:57: PM
ICSE 9 - Chemistry
Asked by enakshipal07 | 19 Nov, 2023, 11:45: AM











