CBSE Class 9 Answered
Calculate the mass in grams for the following: A)0.5 mol of calcium sulphate (caSo4). B) 0.25 mol of carbon dioxide gas (co2) { cu=40,s=32,o=16,c=12}.
Asked by saraswatiupadhaya111 | 26 Aug, 2020, 08:54: AM
Mass in grams for the following can be calculated by calculating number of moles.
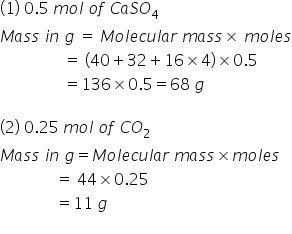
Answered by Ravi | 27 Aug, 2020, 18:03: PM
Application Videos
Concept Videos
CBSE 9 - Chemistry
Asked by gauravsingh36428 | 14 Mar, 2022, 19:44: PM
CBSE 9 - Chemistry
Asked by krishnaahlawat062 | 27 May, 2021, 21:01: PM
CBSE 9 - Chemistry
Asked by kinshukgoel | 12 Sep, 2020, 13:26: PM
CBSE 9 - Chemistry
Asked by saraswatiupadhaya111 | 26 Aug, 2020, 08:54: AM
CBSE 9 - Chemistry
Asked by sonydatta5 | 24 Aug, 2020, 10:18: AM
CBSE 9 - Chemistry
Asked by Minajoshi62 | 02 Feb, 2020, 21:01: PM
CBSE 9 - Chemistry
Asked by tarunvignesh2005 | 12 Jan, 2020, 17:46: PM
CBSE 9 - Chemistry
Asked by Virenpareek3 | 23 Nov, 2019, 16:13: PM
CBSE 9 - Chemistry
Asked by fathimafero | 11 Sep, 2019, 14:00: PM
CBSE 9 - Chemistry
Asked by abhishekverma0300 | 27 Aug, 2019, 00:41: AM









