NEET Class neet Answered
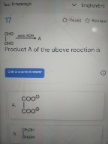
0.17g of ammonia on decomposition gave 0.14g of nitrogen and rest hydrogen . What will be the volume of hydrogen evolved at S.T.P
Asked by Imsneharose | 30 Nov, 2021, 09:34: PM
2NH3 → N2 + 3H2
Moles of NH3 present = 0.17 g x 1 mol/17 g = 0.01 moles
moles H2 produced = 3/2 x 0.01 moles = 0.015 moles H2
At STP 1 mole = 22.4 L
So volume of H2 = 0.015 moles x 22.4 L/mol = 0.336 Liters.
Answered by Ramandeep | 01 Dec, 2021, 06:19: PM
Application Videos
Concept Videos
NEET neet - Chemistry
Asked by dikshitgod07 | 29 Apr, 2024, 08:38: AM
NEET neet - Chemistry
Asked by biswassayan8464 | 21 Apr, 2024, 11:30: AM
NEET neet - Chemistry
Asked by mahendar160786 | 16 Apr, 2024, 09:23: PM
NEET neet - Chemistry
Asked by raomayankup83 | 15 Apr, 2024, 07:46: PM
NEET neet - Chemistry
Asked by muskannawab11 | 14 Apr, 2024, 03:13: PM
NEET neet - Chemistry
Asked by 8239682116rahul | 10 Apr, 2024, 01:48: PM
NEET neet - Chemistry
Asked by tarasingrathod63 | 07 Apr, 2024, 01:07: PM
NEET neet - Chemistry
Asked by fathimahusna6122 | 05 Apr, 2024, 10:25: AM
NEET neet - Chemistry
Asked by vasantagomasi23 | 05 Apr, 2024, 08:35: AM