Class 10 FRANK Solutions Chemistry Chapter 1 - Periodic Properties And Variation Of Properties: Physical And Chemical
Practise Frank Solutions for ICSE Class 10 Chemistry Chapter 1 Periodic Properties and Variation of Properties – Physical and Chemical to understand the modern periodic law. Revise Chemistry concepts such as ionisation potential, electron affinity etc. in this chapter. At our study portal, get accurate answers for short answer questions, true or false questions, MCQs and fill in the blank questions from the Frank textbook.
TopperLearning is a preferred study portal for students who seek quality learning resources to increase their Chemistry marks. Other than ICSE Class 10 Chemistry Frank Solutions, you can browse our Selina Solutions and other chapter resources too.
Periodic Properties And Variation Of Properties: Physical And Chemical Exercise 16
Solution 1
Concept Insight: The elements are characterized by their atomic number as well as atomic weight. Modern periodic law uses atomic number which is number of protons or number of electrons present in an atom of an element.
Solution 2
Concept Insight: Classification of elements on the basis of increasing atomic number is known as Modern Periodic Table. The vertical columns are called groups and the horizontal rows are called periods.
Solution 3
Concept Insight: Periodicity in properties is due to the repetition of similar outer electronic configuration of elements at certain regular intervals.
Solution 4
In general, the elements belonging to a period do not have same valency but their valence shell remains the same. For example, second period has 8 elements with atomic number 3 to 10 but in all of them the valence electrons are present in shell number two.
Concept Insight: For elements in a group the number of electrons present in the outermost shell is the same and therefore the elements have same valency and or elements in a period number of electrons present in the outermost shell of elements in a period increase from left to right but the shell does remains the same.
Solution 5
Concept Insight: For answering this question you should recall that electron affinity is the energy released when an electron is added to an isolated gaseous atom to form an anion. Due to small size of fluorine atom, the valence shell is already crowded, hence when an electron is added to a fluorine atom in gaseous state there occurs strong repulsion between the added electron and those already present in the atom hence less amount of energy is released.
Solution 6
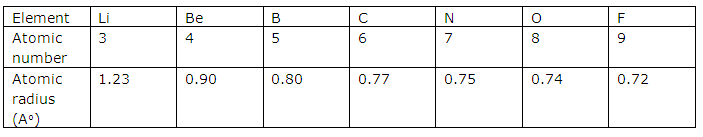
(ii) The element with highest electro negativity: Fluorine (F)
(iii) The element with largest atomic size: Lithium (Li)
(iv) The most reactive non-metal: Fluorine
(v) The most reactive metal: Lithium
Concept Insight: (i) Neon has highest ionization energy since it is a noble gas and has its octet complete which makes it very stable.
(ii) Fluorine has highest electronegativity as we know that electro negativity increases along a period due to decreasing atomic size and increasing nuclear charge.
(iii) Lithium has the largest atomic size since it is an alkali metal i.e., belongs to group 1 and we know that as we move from left to right in a period atomic size decreases. So lithium has largest size while fluorine has smallest size in second period.
(iv) Fluorine is most reactive non metal as it requires only one electron to complete its octet and become stable.
(vi) Lithium is the most reactive metal as it can complete its octet by losing its single electron present in its outermost shell.
Periodic Properties And Variation Of Properties: Physical And Chemical Exercise 17
Solution 7
(ii) The most non-metallic element will be found at D.
Concept Insight: For answering this question you should recall metallic character increases down the group and also increases with the increasing size of the atom. Since elements of group 1 has largest atomic sizes among all the elements of periodic table so the most metallic element belongs to group 1.
Similarly, non-metallic character decreases down the group and increases with the decreasing size of atom.
Solution 8

Solution 9
(i) 18, 7.
(ii) First.
(iii) Seventeen.
(iv) Electron affinity.
(v) Decrease, increase
(vi) Fluorine
(vii) Zero.
Solution 10
(ii) True.
(iii) True.
(iv) True.
(v) True.
(vi) True.
Concept Insight: (i) There is inverse relation between atomic size and electron affinity. More is the size, less is the electron affinity and vice versa because more is the size of atom, more is the distance between the nucleus and last shell to which electron enters. This results in decrease in force of attraction between the nucleus and incoming electron and hence the electron affinity decreases. Fluorine has smaller size than chlorine so it must have less electron affinity than chlorine.
Solution 11
(ii) I < Br < F < Cl
(iii) SiO2 < P2O5 < SO3 < Cl2O7
(iv) I+ < I < I-
Concept Insight:
(i) Li, Be, B belongs to second period and ionization energy increase as we move left to right in a period due to increased nuclear charge and decrease of atomic size.
(ii) Electron affinity decreases down the group due to increase in atomic size as it results in more distance between nucleus and last shell to which incoming electron enters. Hence, incoming electron feels less attraction from the nucleus.
(iii) In a period, acidic nature of oxide increases.
(iv) Size of a cation is always smaller than the corresponding atom due to decrease in number of electrons and increase in effective nuclear charge i.e., greater force of attraction by the nucleus on the electrons.
Size of an anion is always more than the corresponding atom due to decrease in effective nuclear charge i.e., lesser force of attraction by the nucleus o the electrons.
Periodic Properties And Variation Of Properties: Physical And Chemical Exercise 18
Solution 12
The statement that in each period, the atomic size gradually decreases with increase in atomic number means that as move from left to right in a period, nuclear charge increases by one unit in each succeeding element while the number of shells remains the same. Due to this increased nuclear charge, the electrons of all the shells are pulled closer to the nucleus thereby bringing the outer most shell closer to the nucleus. With the result, the atomic size decreases across a period.
For example, in the second period from lithium to fluorine, lithium has the largest size while fluorine has the smallest size.
Solution 13
In the case of noble gases or inert gases there are exceptions and the atomic radius or size of the elements are greater than the other elements of the period to which these elements belong.
Solution 14
Although nuclear charge also increases in going down the group but the effect of nuclear charge on atomic size is much less than the increase due to addition of a new shell.
In group 17, the atomic size follows the trend:
F < Cl < Br < I
Solution 15
Na, Mg, Al, Si, P, S, Cl, Ar
The most metallic element is sodium i.e., Na and the most non-metallic element is chlorine i.e., Cl.
Concept Insight:
In a period, metallic character decreases on moving from left to right because of decrease in size of atom due to which elements cannot lose electron easily.
Solution 16

Solution 17
Solution 18
Unit: Its units are electron volt (eV).
Its SI units are Kilojoules per mole (KJmol-1).
Solution 19
Solution 20
(ii) Halogens need only one electron to complete their octet and become stable their atomic size is very less hence the distance between their last shell and nucleus is very less, as a result the force of attraction between the nucleus and the incoming electron is less and hence the electron affinity is high for halogens.
(iii) When an atom loses or gain electron to form ion, the number of electrons present in the outermost shell also changes. Corresponding to that effective nuclear charge on the changed number of electrons also change which further changes the size of an atom as there is inverse relation between effective nuclear charge and size of atom.
(iv) K and Li belongs to group 1 i.e., metals and we know that for metals chemical reactivity of elements increases down the group because chemical reactivity increases as electropositive or metallic character increases.
(v) The electronegativity of chlorine is higher than sulphur because both of them belong to third group and chlorine follows sulphur. We know that, within a period electronegativity increases as we move from left to right because of decrease in atomic size and increase in nuclear charge.
(vi) Group 17 elements are non metals because they have 7 electrons in their valence shell and ionize by accepting 1 electron to form an anion.
For example group 17 elements F, Cl, Br and I all have 7 electrons in their valence shell and ionize by accepting 1 electron to form F-, Cl-, Br- and I-.
Group 1 elements are metals because they have tendency to lose the one electron present in their valence shell and form positive ion.
For example, group 1 elements Li, Na, K, Rb, Cs have tendency to lose the one electron present in their valence shell and form positive ions Li+, Na+, K+, Rb+ and Cs+.
Solution 21
(i) (C) i.e. 2, 8, 2 because it has only 2 electrons in its valence shell which can be lost to form a di positive cation.
(ii) (C) i.e. 0.72, 0.72
(iii) (d) i.e. element forms basic oxide because the element is a metal as it has valency 1.
(iv) (a) i.e. F because it belongs to group 17 whose elements have valency 7 and thus requires only 1 electron to complete their octet.
Solution 2000-1
In period 2 = 8
In period 3 = 8.
(ii) Elements in period 1 are Hydrogen (H) and Helium (He).
(iii) Atomic size of elements decreases on moving from left to right in a period.
Periodic Properties And Variation Of Properties: Physical And Chemical Exercise 19
Solution 2000-2
(ii) An element in group 7 is likely to be non metallic in character since group 7 element will have 7 electrons in its valence shell.
(iii) Metallic.
Solution 2001-1
2. Period, non-metallic.
3. More.
4. Number of outer electrons.
Solution 2002-1
Solution 2002-2
Solution 2002-3
Solution 2002-4
Solution 2003-1
(ii) Covalent.
(iii) The elements of group VIIA all have same number of electrons in their valence shell and same valency.
(iv) Neon
(v) 8 electrons are present in the valence shell of the element with atomic number 18.
(vi) Electron affinity.
(vii) Electronic configuration of element in the third period which gains one electron to become an anion is 2, 8, 7.
(viii) Decreases, number of valence shell electrons/outermost shell electrons, valence shell/ outermost shell.
Periodic Properties And Variation Of Properties: Physical And Chemical Exercise 20
Solution 2004-1
(ii) (a) lower, higher.
(b) remains the same.
(c) remains the same.
Solution 2005-1
(ii) d
(iii) c
(iv) a
(v) c
Periodic Properties And Variation Of Properties: Physical And Chemical Exercise 21
Solution 2006-1
(b) Nitrogen. It should be placed between oxygen andcarbon.
(c) Beryllium < nitrogen < fluorine
(d) Fluorine (F)
Solution 2007-1
(b) Boron
(c) 3
(d) BCl3
(e) The elements in the group to the right of this boron group will be less metallic in character because on moving to the right of the periodic table metallic character decreases as ionization energy deceases and tendency to lose electron also decreases.
Solution 2008-1
(b) True
(c) False
(d) True
Solution 2008-2
(a) (i) First element in period 2 is Lithium and last element is Neon.
(ii)Atomic size increases on moving from top to bottom of a group.
(iii)Chlorine among halogens has the greatest electron affinity.
(iv) All elements in group 7 have same number of valence shell electrons.
(b) (i) metallic
(ii) smallest
(c) (i) Ba i.e. Barium will form ion most readily since it is at the bottom of a group its ionization energy is low because its atomic size is more. Due to this effective atomic charge of nucleus over the valence shell electron is least and it can be removed easily.
(ii) Electro negativity of an element measures the capacity of an element to attract the shared pair of electrons in a bond towards itself.
Periodic Properties And Variation Of Properties: Physical And Chemical Exercise 22
Solution 2009-1
Solution 2009-2
Solution 2010-1
Atomic number of an element is 16.
- It belongs to Period 16.
- Number of valence electrons in the element is 6.
- The element is a non-metal.
Solution 2011-1
The oxidising power of elements depends on the tendency to gain electrons which increases from left to right along a period due to increase in nuclear pull.
Solution 2011-2
Three shells indicate that the element belongs to the third period.
Three valence electrons indicate that the element belongs to the third group.
Solution 2011-3
- Across a period, the ionisation potential increases.
- Down the group, electron affinity decreases.
Periodic Properties And Variation Of Properties: Physical And Chemical Exercise 23
Solution 2011-4
(i) In the periodic table, alkali metals are placed in Group I. So, the correct option is A.
(ii) The correct option is C.
The elements of halogen family are non-metallic in nature.
Solution 2013-1
Correct option: (D) Fluorine
Solution 2013-2
(c) Zinc oxide
Solution 2013-3
The element which has the highest ionisation potential is helium (He).
Solution 2013-4
- I
- R
- M
- 5
- T
- Y
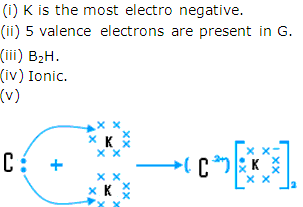
- Ionic bond will be formed and the molecular formula is A2H.
Solution 2014-1
- Correct option: D (atomic radius decreases and nuclear charge increases)
Solution 2014-2
- Correct option: A (3 shells and 2 valence electrons)
Periodic Properties And Variation Of Properties: Physical And Chemical Exercise 24
Solution 2014-3
(a) An element Z having atomic number 16 is Sulphur.
(i) Sulphur belongs to Period 3 and Group 16.
(ii) Sulphur is a non-metal.
(b) Two hydrogen atoms combine with one sulphur atom to form hydrogen sulphide (H2S) gas.
Solution 2015-1
(i) Lithium
Reason: Electronegativity increases from left to right. Lithium is present on the left side of the periodic table; hence, it will be the least electronegative element.
Solution 2015-2
(1) Ba metal will form ions readily because the ionsation energy decreases down the group as the size increases.
(2) On moving down the group, the number of electrons in the outermost shell, i.e. valence electrons, remain the same. So, the valency in the group remains the same.
Solution 2015-3
a. Mg, Cl, Na, S, Si (increasing order of atomic size) -
Cl < S < Si < Mg < Na
99 pm < 104 pm < 117 pm < 160 pm < 186 pm
b. Cs, Na, Li, K, Rb (increasing metallic character)
Li < Na < K < Rb < Cs
c. Na, K, Cl, S, Si (increasing ionisation potential) -
Cl < S < Si < Na < K
1256 < 999 < 786 < 496 < 419
d. Cl, F, Br, I (increasing electron affinity) -
I < Br < F < Cl
-295 KJ mol-1 < -324 KJ mol-1 < -327.9 KJ mol-1 < -349 KJ mol-1
e. Cs, Na, Li, K, Rb (decreasing electronegativity) -
Li > Na > K = Rb > Cs
1.0 > 0.9 > 0.8 = 0.8 > 0.7
Solution 2015-4
(a) A basic oxide: Na2O (Na2O + H2O → 2NaOH)
(b) An oxide which dissolves in water forming an acid: SO2 (SO2+ H2O → H2SO3)
(c) An amphoteric oxide: Al2O3 (shows both acidic and basic properties)
(d) A covalent oxide of a metalloid: SiO2 (Si is a metalloid)
Solution 2016-1
(i) < (less than)
(ii) < (less than)
Solution 2016-2
(a) The element J is placed in the 5th group; hence, its number of valence electrons is 5.
(b) The element M which is placed in the 7th group has 7 valence electrons and will gain only one electron to complete its octet; hence, element M will show a single negative charge.
(c) Element T is more reactive than element R because chemical reactivity increases down the group.
(d) Only element T is placed in the 4th period; hence, its electrons are arranged in four shells.
Periodic Properties And Variation Of Properties: Physical And Chemical Exercise 25
Solution 2016-3
(a) If an element has low ionisation energy, then it is likely to be metallic.
(b) If an element has seven electrons in its outermost shell, then it is likely to have the smallest atomic size among all the elements in the same period.
Solution 2017-1
(a) The energy required to remove on electron from a natural isolated gaseous atom and convert it into a positively charged gaseous ion is called ionisation potential.
Solution 2017-2
(a) He < Ne < Ar
(b) K < Na < Li
(c) Br < Cl < F
(d) Li < Na < K
Solution 2017-3
(a) A solid non-metal belonging to the third period: 15
(b) A metal of valency 1: 19
(c) A gaseous element with valency 2: 8
(d) An element belonging to Group 2: 4
(e) A rare gas: 2