Class 9 SELINA Solutions Chemistry Chapter 4: Atomic Structure and Chemical Bonding
Atomic Structure and Chemical Bonding Exercise Ex. 4(A)
Solution 1
a. According to MaharshiKanada:
i. Matter consisted of indestructible particles called paramanus (now called atoms).
ii. A paramanu does not exist in the free state, rather it combines with other paramanus to form a bigger particle called the anu (now called a molecule).
b. Democritus called paramanu as atom which comes from the Greek word atomos, meaning indivisible.
Solution 2
Main postulates of Dalton's atomic theory:
i. Matter consists of very small and indivisible particles called atoms.
ii. Atoms can neither be created nor be destroyed.
iii. The atoms of an element are alike in all respects, but they differ from the atoms of other elements.
iv. Atoms of an element combine in small numbers to form molecules.
v. Atoms of one element combine with atoms of another element in a simple ratio to form molecules of compounds.
vi. Atoms are the smallest units of matter which can take part in a chemical reaction.
Solution 3
An α-particle is a doubly charged helium ion (He2+) containing two protons and two neutrons. It is formed by removing two electrons from the helium atom.
Solution 4
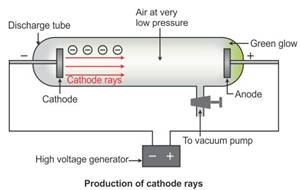
Cathode rays are the beam of electrons which travel from the negatively charged end (i.e. cathode to anode) of a vacuum tube, across a voltage difference between the electrodes placed at each end.
Formation of cathode rays:
Generally gases are poor conductors of electricity. However, when a high voltage charge from an induction coil is applied to tubes filled with gases at very low pressure (0.01 mm of mercury), the gases become good conductors of electricity and begin to flow in the form of rays.
These rays are called cathode rays and they travel from the cathode towards the anode.
Solution 5
- Cathode rays are negatively charged as they consist of negatively charged particles called electrons.
- Anode rays are made of positively charged particles.
Solution 6
When a beam of cathode rays is made to fall upon hard metallic targets like tungsten, X-rays are produced.
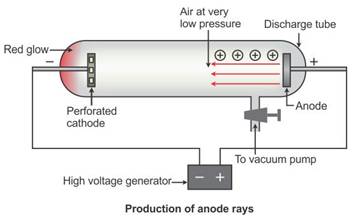
Solution 7
When perforated rays were used in the discharge tube, another set of rays travelling in a direction opposite to that of the cathode rays, i.e. from the anode towards the cathode, is seen, as in the figure below. These rays are called canal rays because they passed through the holes or canals in the cathode. These rays were named positive rays or anode rays.
Solution 8
|
Cathode rays |
Anode rays |
|
These rays travel from the cathode to the anode. |
These rays travel from the anode to the cathode. |
|
They are made of negatively charged particles. |
They are made of positively charged particles. |
|
They produce a greenish yellow fluorescence on a soda-glass screen. |
They produce fluorescence on a zinc sulphide screen. |
|
They are affected by an electric field, i.e. they are inflected towards a positive field and deflected towards a negative field. |
They are deflected by electric and magnetic fields but in a direction opposite to that of cathode rays. |
Solution 9
Chadwick discovered neutral particles present in an atom by bombarding light nuclei like beryllium with alpha particles, i.e. helium nuclei.
4Be9 + 2He4 → 6C12 + 0n1
Discovery of the neutron also showed that an atom is divisible.
Solution 10
a. Hydrogen does not contain any neutron.
b. No. It is neutral.
Solution 11
Thomson's Atomic Model:
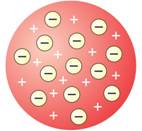
According to Thomson's atomic model, an atom is made of positively charged substances in the form of a sphere. Electrons are embedded into this sphere, and the total positive charge of the sphere is equal to the total negative charge of electrons, and hence, the atom remained electrically neutral. This model was not accepted because it was unable to explain how positively charged particles were shielded from negatively charged particles without getting neutralised.
|
|
|
|
Thomson's atomic model |
Plum pudding |
Solution 12
a. Thomson discovered particles.
b. Goldstein discovered protons.
c. Chadwick discovered neutrons.
Solution 13
a. Proton
b. Electron
c. Neutron
Solution 14(a)
Rutherford used gold for his scattering experiment because gold is the most malleable metal and he wanted the thinnest layer as possible.
The gold sheet used was around 1000 atoms thick.
Therefore, Rutherford selected a Gold foil in his alpha scattering experiment.
Solution 14(b)
- If the foil of a heavy metal like platinum (having a very heavy nucleus) is used, then the observations in the α-particle scattering experiment would be the same as that in the gold foil experiment
- If, however, the foil of a right metal like lithium (having a very light nucleus) is used, then the fast moving heavy α-particles may even push the light nucleus aside and may not be deflected back.
Solution 15
According to Rutherford's model of an atom, the subatomic particle in a nucleus is a proton.
Solution 16
The nucleus was discovered by Rutherford.
Solution 17
Rutherford performed an experiment by allowing a stream of alpha particles to pass through a very thin gold foil. He observed that alpha particles pass through the metal foil without deviating from their path. This shows that an atom contains a large empty space called nuclear space.
Solution 18
One major drawback of Rutherford's model was the comparison of electrons with the planets in the solar system.
Thus, when an electron moved around the nucleus continually, it should radiate energy, i.e. lose energy. As a result it should be gradually pulled towards the nucleus and end up colliding with it. This should result in the total collapse of the atom.
However, we know that the atom is structurally stable. Thus, Rutherford's model could not explain this stability.
Solution 19
a) A is for K shell or I shell.
B is for L shell or II shell.
C is for M shell or III shell.
Shell K has the minimum amount of energy.
b) X is a nucleus; it is positively charged.
c) The above sketch is of Bohr model of an atom.
Solution 20
- Atom has a central nucleus surrounded by electrons.
- The electrons revolve around the nucleus in definite circular paths called orbits.
- Each orbit has fixed energy. Therefore, these orbits are also known as energy levels or energy shells.
- These orbits or energy levels are represented either by a number 1, 2, 3,... known as the principal quantum number (n) of the orbit or by K, L, M,....
- There is no change of energy of electrons as long as they keep revolving in the same energy level and the atom remains stable. Electrons can jump from the higher orbit to the lower orbit when they lose energy and jump to the next when they gain energy.
Atomic Structure and Chemical Bonding Exercise Ex. 4(B)
Solution 1
- The three fundamental particles of an atom are electron proton and neutron.
|
Particle |
Symbol |
Charge |
|
Electron |
e |
-1 |
|
Proton |
p |
+1 |
|
Neutron |
n |
No charge |
Solution 2
|
Element |
Symbol |
No. of Protons |
No. of Neutrons |
No. of Electrons |
|
Sodium |
|
11 |
12 |
11 |
|
Chlorine |
|
17 |
18 |
17 |
|
Uranium |
238U92 |
92 |
146 |
92 |
|
S |
|
9 |
10 |
9 |
Solution 3
Atomic number = Number of protons or number of electrons = 4
Mass number = Number of protons + Number of neutrons = 4 + 5 = 9
Solution 4
Atomic number (11) of sodium conveys information that the number of protons and electrons is the same.
Mass number (23) of sodium indicates the sum of protons and neutrons.
Solution 5
p = proton
n = neutron
e = electron
Superscript number : These superscript number show their mass number
Subscript number : These numbers show their atomic number
Solution 6
Mass number = 24
Atomic number = 12
No. of electrons = 24 - 12 = 12
Electronic configuration = 2, 8, 2
Solution 7
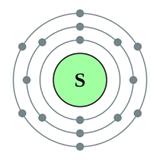
Atomic number = 16
Atomic mass = 32
Number of protons = 16
Number of electrons = 16
Number of neutrons = 32 - 16 = 16
Electronic configuration = 2, 8, 6
Solution 8
i. The maximum capacity of a shell to accommodate electrons is given by the general formula 2n2, where n is the serial number of a shell.
ii. The maximum number of electrons possible in the outermost shell is 8 and that in the penultimate shell is 18.
iii. It is not necessary for an orbit to be completed before another is formed. In fact, a new orbit is formed when the outermost shell attains 8 electrons.
Solution 9
Orbital diagram of ![]()
Atomic number of Ca2+ is 18.
Electronic configuration of Ca2+ is 2, 8, 8.
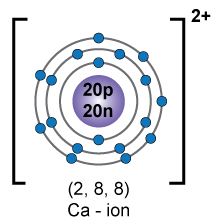
Number of three fundamental particles of Ca2+:
Protons: 18
Electrons: 18
Neutrons: 40 - 18 = 22
Solution 10
Electronic configuration: 27X13 = 2, 8, 3
Number of electrons in X = 13
Number of neutrons in X = 27 - 13 = 14
Electronic configuration of 35Y17 = 2, 8, 7
Number of electrons in Y = 17
Number of neutrons in Y = 35 - 17 = 18
Hence, formula 27X13 stands for 27Na13 and 35Y17 for 35Cl17
Compound formula = NaCl
Atomic Structure and Chemical Bonding Exercise Ex. 4(C)
Solution 1. a
Correct option: (iii) Atomic nucleus
Solution 1. b
Correct option: (i) [6]
Solution 1. c
Correct option: (iii) [2, 8, 8, 1]
Solution 1. d
Correct option: (i) 23
Solution 1. e
Correct option: (iv) 18
Solution 1. f
Correct option: (iv) - 2, 7
Solution 1. g
Correct option: (iii) - ![]()
Solution 1. h
Correct option: (iii) - 1
Solution 1. i
Correct option: (iii) Can be metals or non-metals
Solution 1. j
Correct option: (ii) - Protons and neutrons
Solution 1. k
Correct option: (ii) - 13
Solution 1. l
Correct option: (i) - 18
Solution 1. m
Correct option: (i) - K
Solution 1. n
Correct option: (ii) - Noble gas
Solution 1. o
Correct option: (iii) - Neutrons
Solution 2
a. Hydrogen
b. Helium
c, Magnesium
d. Hydrogen and carbon
e. K
Solution 3
|
Atomic Number |
Name with valency |
| a. 15 |
A solid non-metal of valency 3 |
| b. 8 |
A gas of valency 2 |
| c. 19 |
A metal of valency 1 |
| d. 14 |
A non-metal of valency 4 |
Solution 4
a. The elements have a complete outermost shell, i.e. 2 or 8 electrons. They ordinarily do not enter into any reaction.
b. These exist as monoatoms because molecules of these elements contain only one atom.
c. Valence electrons: The number of electrons present in the valence shell is known as valence electrons.
Solution 5
The three isotopes differ only due to their mass number which is respectively 1, 2 and 3 and named protium, deuterium and tritium.
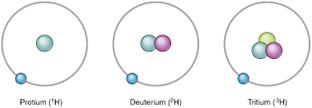
Solution 6
The number of protons in the nucleus of an atom determines the element type of the atom.
Solution 7
a. X1+
b. Oxidising agent, because it has the ability to donate electrons.
Solution 8
a. Mass number is the sum of the number of protons and the number of neutrons in the nucleus of an atom.
b. Ion is an atom or molecule which carries a positive or negative charge because of loss or gain of electrons.
c. Cation is a positively charged ion which is formed when an atom loses one or more electrons; for example, Na+, Hg2+ and Ca2+.
d. Anion is a negatively charged ion which is formed when an atom gains one or more electrons; for example, Cl- and I-.
e. Element is a substance which cannot be split up into two or more simple substances by usual chemical methods of applying heat, light or electric energy; for example, hydrogen, oxygen and chlorine.
f. Orbit is a circular path around the nucleus in which electrons of the atom revolve.
Solution 9
Atomic number = 2
Mass number = 4
Solution 10
Number of electrons in the M shell = 2
So, the number of electrons in the K and L shells will be = 2, 8
Hence, atomic number = 2 + 8 + 2 = 12
Number of protons = 12
Solution 11
a.
i. Atom E contains 7 protons.
ii. Atom B has an electronic configuration 2, 7.
b. Atom C stands for 7Li3, Atom D stands for 8O16.
Hence, compound formula is Li2O.
c. Metals: A and C, Non-metals: B, D, E
Solution 12
i.
|
|
12Mg24 |
12Mg26 |
|
No. of electrons |
12 |
12 |
|
No. of protons |
12 |
12 |
|
No. of neutrons |
24 - 12 = 12 |
26 - 12 = 14 |
Hence, composition of nuclei
12Mg24 → 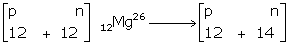
ii. Electronic configuration = 2, 8, 2
- Mass numbers of two isotopes of magnesium are different because of different number of neutrons, i.e. 12 and 14, respectively.
Solution 13
Nucleons: Particles which constitute the nucleus are called nucleons.
Protons and neutrons are the nucleons.
Atomic mass of phosphorus = 31
Atomic number = 15
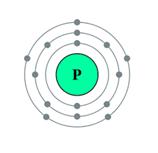
Solution 14
Isotopes: Atoms of the same element having the same atomic number but different mass numbers.
Isotopes differ with reference to neutrons.
Uses of isotopes:
i. Some isotopes are radioactive, i.e. isotopes of cobalt are used for treating cancer and other diseases.
ii. An isotope of 235U is used as a fuel in a nuclear reactor.
Solution 15
Only electrons take part in chemical reactions. Chemical properties depend on the electronic configuration. Isotopes of the element Cl ![]() have the same atomic number, and hence, the same configuration. So, they have the same chemical properties. These differ only in physical contents and weights because neutrons contribute to the mass of an atom.
have the same atomic number, and hence, the same configuration. So, they have the same chemical properties. These differ only in physical contents and weights because neutrons contribute to the mass of an atom. ![]() have different number of neutrons 18 and 20, respectively.
have different number of neutrons 18 and 20, respectively.
Solution 16
Atomic masses of the isotopes of chlorine are 35 and 37. However, in any given sample of chlorine gas, the isotopes occur in the approximate ratio 3:1, 75% of Cl35 and 25% of Cl37. So, the relative atomic mass or atomic weight of chlorine is 35.5.
Fractional atomic mass of chlorine
At. Mass = 3(35) + 1(37) = 105 + 37
2 2
Solution 17
a. Atomic number of an element is the number of protons in the nucleus of an atom.
b.
|
|
No. of protons |
No. of electrons |
No. of neutrons |
Atomic number |
Mass number |
|
|
17 |
17 |
18 |
17 |
35 |
|
|
17 |
17 |
20 |
17 |
37 |
c.
i. Electronic configuration of chlorine = 2, 8, 7
ii. Electronic configuration of chloride ion = 2, 8, 8
Solution 18
- Sodium atom
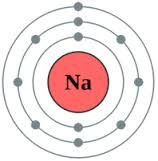
- Chloride ion
- Carbon atom
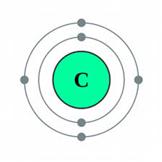
- Oxide ion
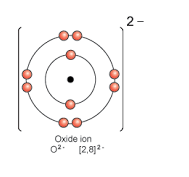
Solution 19
|
Element Symbol |
Atomic Number |
Mass Number |
Numbers of Neutrons |
Number of Electrons |
Number of Protons |
|
Li |
3 |
6 |
4 |
3 |
3 |
|
Cl |
17 |
37 |
20 |
17 |
17 |
|
Na |
11 |
23 |
12 |
11 |
11 |
|
Al |
13 |
27 |
14 |
13 |
13 |
|
S |
16 |
32 |
16 |
16 |
16 |
Solution 20
a. Physical properties depend on atomic mass, and isotopes have different mass number, i.e. they have different number of neutrons. So, isotopes have different physical properties.
b. Argon does not react as it has the outermost orbit complete, i.e. 8 electrons in the outermost shell.
c. Actual atomic mass is greater than the mass number because the mass number is a whole number approximation of atomic mass unit. In fact, neutrons are slightly heavier than protons, and an atom has over 200 sub-atomic particles.
d. ![]() are isotopes of chlorine element which differ in the number of neutrons, whereas chemical properties are determined by the electronic configuration of an atom. Isotopes of an element are chemically alike.
are isotopes of chlorine element which differ in the number of neutrons, whereas chemical properties are determined by the electronic configuration of an atom. Isotopes of an element are chemically alike.
Solution 21
The use of the following isotopes:
(a) Radioactive isotope - used in industry to detect the leakage in understand oil pipelines, gas pipelines and water pipes.
(b) ![]() - Used as a fuel in nuclear reactors.
- Used as a fuel in nuclear reactors.
(c) ![]() - Used in radiotherapy for treating cancer
and other diseases.
- Used in radiotherapy for treating cancer
and other diseases.
(d) ![]() - Used in the
treatment of goitre
- Used in the
treatment of goitre
(e) ![]() - used for
determining the age of historical and geological material.
- used for
determining the age of historical and geological material.
Solution 22
The latest research on the atom has proved that most of the postulates of Dalton's atomic theory contradict. However, Dalton was right that atoms take part in chemical reactions.
Comparisons of Dalton's atomic theory with the modern atomic theory.
Dalton's atomic theory:
i. Atoms are indivisible.
ii. Atoms of the same element are similar in every respect.
iii. Atoms combine in a simple whole number ratio to form molecules.
iv. Atoms of different elements are different.
v. Atoms can neither be created nor be destroyed.
Modern atomic theory:
i. Atoms are no longer indivisible and consist of electrons, protons, neutrons and even more sub-particles.
ii. Atoms of the same element may differ from one another called isotopes.
iii. Atoms of different elements may be similar called isobars.
iv. Atoms combine in a ratio which is not a simple whole number ratio; e.g. in sugar, the C12H22O11 ratio is not a whole number ratio.
Solution 23. a
Elements tend to combine with one another to attain the stable electronic configuration of the nearest inert gas, i.e. 8 electrons in its valence shell. This is known as the Octet Rule.
Solution 23. b
Hydrogen
Hydrogen atom has one electron in the valence shell which it shares with another hydrogen atom having one electron to complete its duplet state, i.e. two electrons in the valence shell and resulting in the formation of hydrogen.
Solution 24
(i)
Element A
Atomic number = 7 = Number of electrons = 2, 5
Valency of A = 8 - 5 = 3
Element B
Electronic configuration 2, 8, 8
Valency of B = Zero
Element C has 13 electrons
Electronic configuration = 2, 8, 3
Valency of C = 3
Element D
Protons = 18 = Electrons = 2, 8, 8
Valency of D = Zero
Element E
Electronic configuration = 2, 8, 8, 1
Valency of E = 1
(ii) C and E are metals.
(iii) A is a non-metal.
(iv) A, C and E are not inert gases.
Solution 25
Atomic numbers of X Y Z
6 9 12
(2, 4) (2, 7) (2, 8, 2)
a. Y(2, 7) forms an anion.
b. Z(2, 8, 2) forms a cation.
c. X(2, 4) has four electrons in the valence shell.
Atomic Structure and Chemical Bonding Exercise Ex. 4(D)
Solution 1
Combining atoms attain the noble gas configuration by transferring or sharing electrons.
Solution 2
The chemical bond formed due to the electrostatic force of attraction between a cation and an anion is called an electrovalent bond.
Solution 3
Metals have a tendency to lose their valence electrons (1, 2 or 3). So, they combine with non-metals which have 7, 6 or 5 electrons in their valence shell to form an electrovalent bond.
Solution 4
- An atom X will lose its three electrons to acquire the noble gas configuration and form a positive ion (a cation).
- The formulae are
i. X(SO4)3
ii. X(NO3)3
iii. XPO4
iv. X2(CO3)3
v. X(OH)3
Solution 5
Besides chaos, everything in this world wants stability. The same is the case with atoms. For atoms, stability means having the electron arrangement of an inert gas, i.e. octet in the outermost shell. Helium has two electrons (duplet), while all other inert gases, i.e. neon, argon, krypton, xenon and radon have eight electrons (octet) in the outermost shell.
Solution 6
Electrovalent compounds are usually formed between metals and non-metals.
Atoms of metallic elements which have 1, 2 or 3 valence electrons can lose electron(s) to atoms of non-metallic elements which have 5, 6 or 7 valence electrons and thereby form an electrovalent bond.
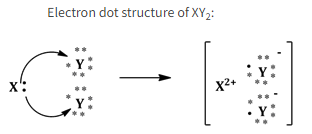
Solution 7. a
Nature of bond in XY2 is an ionic bond.
Solution 7. b
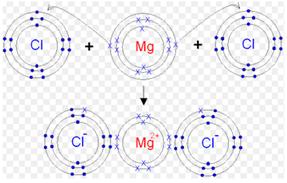
The orbit structure of XY2 :
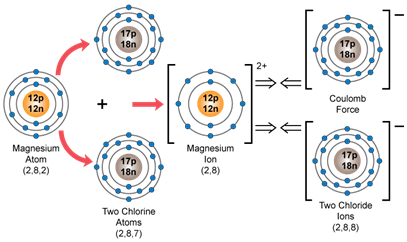
Suppose X is magnesium metal and Y is chlorine. Hence, the orbital diagram for XY2, i.e. for MgCl2 would be as shown below:
Solution 8
a. Ionic bond
b. XY
Solution 9
NaCl
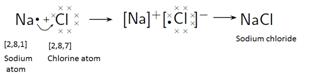
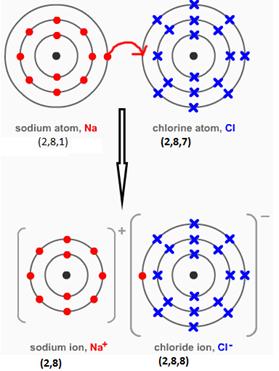
MgCl2
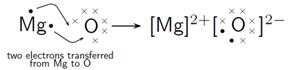
CaO
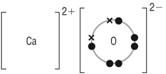
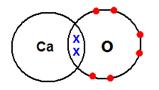
Solution 10
a.
|
Sodium atom |
Sodium ion |
|
Sodium atom is electrically neutral. |
Sodium ion is positively charged. |
|
In sodium atom, there are 11 protons and 11 electrons, i.e. equal number of protons and electrons. |
In sodium ion, there are 11 protons but 10 electrons, i.e. sodium ion contains lesser number of electrons. |
|
Sodium atom has only one electron in its valence shell. |
Sodium ion has 8 electrons in its valence shell. |
|
Size of a sodium atom is larger than a sodium ion. |
Size of a sodium ion is smaller than a sodium atom. |
b.
i. In chlorine atom, the number of protons (17) is equal to the number of electrons (17).
In chloride ion, there are 17 protons but 18 electrons.
ii. Chlorine atom is electrically neutral. Chloride ion is negatively charged.
iii. Chlorine atom is reactive. It reacts with sodium vigorously forming sodium chloride. Chloride ion is unreactive. It does not react with sodium.
iv. Chlorine (Cl2) is a poisonous, toxic, corrosive gas which is used in the manufacture of bleaching agents and disinfectants. It is non-toxic and readily adsorbed by plants.
Solution 11
Fluoride ion is a negatively charged ion with 9 protons and 10 electrons.
Neon atom is electrically neutral with 10 protons and 10 electrons.
Solution 12
Reduction: In the electronic concept, reduction is a process in which an atom or ion accepts electron(s).
Cu2+ + 2e- → Cu
Solution 13
2K + Cl2 → 2KCl
a. Oxidation: In the electronic concept, oxidation is a process in which an atom or ion loses electron(s).
K → K+ + e-
b. Reduction: In the electronic concept, reduction is a process in which an atom or ion accepts electron(s).
Cl2 + 2e-→ 2Cl-
c. Oxidising agent
An oxidising agent oxidises other substances either by accepting electrons or by providing oxygen or an electronegative ion, or by removing hydrogen or an electropositive ion.
Cl2 + 2e-→ 2Cl-
d. Reducing agent
A reducing agent reduces other substances either by providing electrons or by providing hydrogen or an electropositive ion, or by removing oxygen or an electronegative ion.
K → K+ + e-
Atomic Structure and Chemical Bonding Exercise Ex. 4(E)
Solution A. 1
Correct option: (ii) - Electrovalent
Solution A. 2
Correct option: (iii) - CsF
Solution A. 3
Correct option: (i) - 2, 8, 8, 2
Solution A. 4
Correct option: (iv) - Redox reaction
Solution A. 5
Correct option: (iv) - Solids
Solution A. 6
Correct option: (iii) - Oxygen molecule
Solution A. 7
Correct option: (iii) - Hydrogen chloride
Solution A. 8
Correct option: (iii) - 5
Solution A. 9
Correct option: (iv) - Nitrogen
Solution A. 10
Correct option: (iii) - Neon and Argon
Solution A. 11
b. They are formed between metals and non-metals.
Solution A. 12
Correct option: (i) - It loses electrons and is oxidized.
Solution A. 13
Correct option: (i) - XY2
Solution B. 1
a. Atomic number 15
Electronic configuration (15): 2,8,5
b. Atomic number 19
Electronic configuration (19): 2,8,8,1
c. Atomic number 8
Electronic configuration (8): 2,6
d. Atomic number 10
Electronic configuration (10): 2,8
Solution B. 2
i. Reduction
ii. Oxidation
iii. Reduction
iv. Oxidation
Solution B. 3
(a) Covalent bonding
(b) Electrovalent or ionic bonding
Solution B. 4
(a) Helium
(b) Lithium
(c) Hydrogen: 11H, 21H, 31H
(d) 4018Ar and 4020Ca
(e) Ionic bond
(f) Anion
Solution C. 1
a. The chemical bond formed between two combining atoms by mutual sharing of one or more pairs of electrons is called a covalent bond.
b. Covalent bond formed by
i. Bond formed between two Cl atoms; Cl‒Cl
ii. Bond formed between hydrogen atom and chlorine atom; H‒Cl
Solution C. 2
Element A with 1 electron in its first shell is hydrogen, and element B with 7 electrons in its third shell is chlorine.
So, a single covalent bond is formed between hydrogen and chlorine by sharing one pair of electrons.
Solution C. 3
(a) Y will have negative charge.
(b) Y is getting reduced by addition of electrons.
Solution C. 4
i. Y = 9
ii. Z = 12
- Ionic bond with molecular formula ZY2.
Solution C. 5
Potassium chloride is an electrovalent compound and conducts electricity in the molten or aqueous state because the electrostatic forces of attraction weaken in the fused state or in aqueous solution.
Polar covalent compounds like hydrogen chloride ionise in their solutions and can act as an electrolyte. So, both can conduct electricity in their aqueous solutions.
Solution C. 6
HCl and NH3
Solution C. 7
Formula of compound when combined with sulphur - MS
Formula of compound when combined with chlorine - MCl2
Solution C. 8
a. Polar covalent bond
b. Ionic bond
c. Polar covalent bond
Solution C. 9
(a)
(i) Isotopes: A and B
(ii) Isobars: X and Y
(b)
(i) Isotopes: ![]()
(ii) Isobars: ![]()
Solution D. 1
A single covalent bond is formed by sharing of one pair of electrons between atoms, each atom contributing one electron.
A double bond is formed by sharing of two pairs of electrons between two atoms.
A triple bond is formed by sharing of three pairs of electrons between two atoms.
Solution D. 2
|
MgCl2 - Electrovalent compound |
CCl4 - Covalent compound |
|
They are hard crystalline solids consisting of ions. |
These are gases or liquids or soft solids. |
|
They have high melting and boiling points. |
They have low melting and boiling points. |
|
They conduct electricity in the fused or aqueous state. |
They do not conduct electricity in the solid, molten or aqueous state. |
|
These are soluble in inorganic solvents but insoluble in organic solvents. |
These are insoluble in water but dissolve in organic solvents. |
Solution D. 3
Orbital Diagram:
a) Magnesium chloride
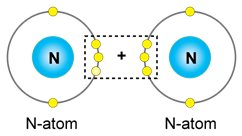
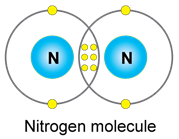
b) Nitrogen
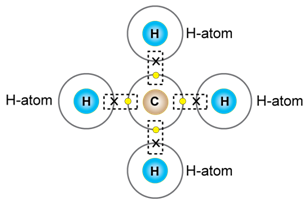
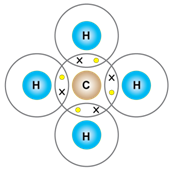
c) Methane
d) Hydrogen chloride
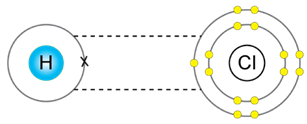
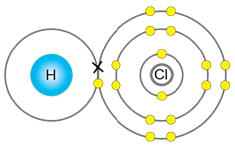
Solution D. 4
The bond formed between metal and nonmetal is ionic bond.
Bond formed between metal M and chlorine is ionic bond.
When metal M combines with sulphur – MgS
When metal M combines with oxygen – MgO
When metal M combines with nitrogen – Mg3N2.
Solution D. 5
- Mass of an atom is contributed by mass of protons and mass of neutrons present inside the nucleus of an atom. Electrons present outside the nucleus are of negligible mass. Therefore, the mass of an atom is concentrated inside the nucleus of an atom.
- Atoms combine because they have incomplete valence shells and tend to attain a stable electronic configuration. So, to attain a stable electronic configuration, atoms of elements gain or share their electrons with other atoms.
- Mass of an atom is contributed by mass of protons and mass of neutrons present inside the nucleus of an atom. Atoms of the same element contain the same number of protons but may differ in their number of neutrons. Therefore, an element has atoms with different mass number.
- Carbon-12 and carbon-14 are isotopes of carbon. They have the same atomic number (6) and hence the same electronic configuration. Chemical properties are determined by the electronic configuration of an atom. So, carbon-12 and carbon-14 show similar chemical properties.
Solution D. 6
- Ethene molecule has one double covalent bond and four single covalent bonds.
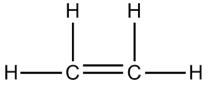
- Ethyne molecule has one triple covalent bond and two single covalent bonds.
![]()
Solution E. 1
(a) Electronic configuration of element A with 6 electrons in its M shell:
2, 8, 6
(b) Atomic number of element A = 16
(c) It is a non-metal.
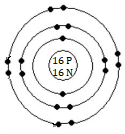
(d) A can form anion. Symbol of the anion = A⁻
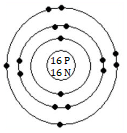
(e) The element A is Sulphur.
(f) The orbital structure of A atom (total 16 electrons, valence electrons = 6)
The orbital structure of A⁻ ion (total 17 electrons, valence electrons = 8)
Solution E. 2
(a) The figure 9 indicates total number of nucleons (i.e. total number of protons + total number of neutrons) present in the nucleus of an atom of Y element.
(b) The figure 4 indicates total number of electrons present in the atomic orbitals of an atom of Y element.
(c) The number of protons in atom Y is equal to number of electrons = 4
(d) What is the number of neutrons in atom Y?
(e) What is the number of electrons in atom Y?
(f) How many electrons are there in the outermost shell of an atom of element Y?
(g) Write the symbol of ion formed by an atom of element Y?
Solution E. 3
Atom A is represented as, ![]() , atom B as
, atom B as ![]() and atom C as
and atom C as ![]() .
.
(a) Electrons, protons and neutrons do A, B and C have
|
|
Electrons |
Protons |
Neutrons |
|
|
82 |
82 |
127 |
|
|
83 |
83 |
126 |
|
|
82 |
82 |
129 |
(b) Two atoms which have different number of nucleons - A and C
(c) The term which can be used for A and C atoms - Isotopes
(d) Isotopes may be defined as atoms of the same element having the same atomic number but different mass number,
Solution E. 4
a. Cation and anion
b. By mutual sharing of electrons
c. Two
d. Magnesium is oxidised and chlorine is reduced.
Solution E. 5
Element X has 2 electrons in its M shell.
The electronic configuration would be 2, 8, 2.
So, element X is Mg (12).
Element Y has 7 electrons in its third orbit.
The electronic configuration would be 2, 8, 7.
So, element Y is Cl (17).
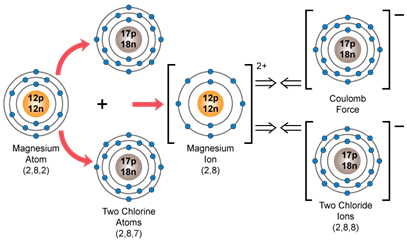
a. MgCl2
b. The nearest inert gas electronic configuration for element X is 2,8, while that for element Y is 2,8,8.
c. Orbital diagram showing the formation of the compound between X and Y:
Magnesium chloride:
Solution E. 6
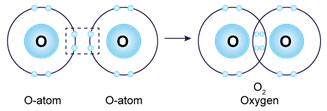
In the formation of
i. Oxygen molecule
a. Neon (10) 2,8
b. Two pairs of electrons are shared.
c. Covalent bond
d. Orbital Diagram:
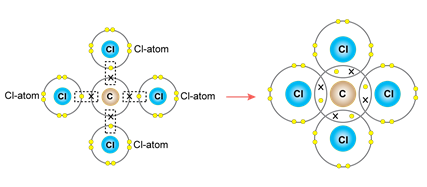
ii. Carbon tetrachloride molecule
a. Neon (10) 2,8
b. Four pair of electrons are shared.
c. Covalent bond
d. Orbital Diagram:
Solution F. 1
The isotopes of bromine exist in about 1:1 ratio.
Hence, average atomic mass of bromine =
![]()
Solution F. 2
Let the % of ![]() be x%. Then the percentage of
be x%. Then the percentage of ![]() be (100 - x) %.
be (100 - x) %.
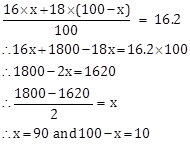
Therefore, % of ![]() is 90 % and 5 of
is 90 % and 5 of ![]() is 10%.
is 10%.