Class 9 FRANK Solutions Physics Chapter 5: Heat - Thermal Expansion
Get free access to complete Frank Solutions for ICSE Class 9 Physics Chapter 5 Heat - Thermal Expansion at TopperLearning. Dive deeper into the concept of heat by learning about the different types of thermal expansion. Also, revise the uses of a thermostat and a bimetallic strip.
Other than the Frank solutions, watch our ICSE Class 9 Physics videos to thoroughly understand the topic of thermal expansion of solids, liquids and gases. If you have trouble understanding the Chapter 5 concepts, post your queries in our ‘UnDoubt’ section for response from our experts.
202
203
Heat - Thermal Expansion Exercise 202
Solution 1
(i). Coefficient of Linear expansion is equal to the change in length of a rod of length 1m when its temperature rises by 10c.
(ii).Coefficient of superficial expansion is equal to the change in area of a rod of area 1m2 when its temperature rises by 10c.
(iii).Coefficient of volume expansion is equal to the change in volume of a rod of volume 1m3 when its temperature rises by 10c.
(ii).Coefficient of superficial expansion is equal to the change in area of a rod of area 1m2 when its temperature rises by 10c.
(iii).Coefficient of volume expansion is equal to the change in volume of a rod of volume 1m3 when its temperature rises by 10c.
Solution 2

Solution 3
A bimetallic strip consists of two metal strips- one with high coefficient of expansion and the other with low coefficient of expansion. Two different metals are used for regulating temperature in an electrical device as the strip bends due to different coefficient of expansion on excessive heating due to current, thus breaking electrical circuit until the strip cools down to a preset point. Two applications of bimetallic strip are thermostat in electric iron and in balance wheels.
(a). When boiling water is poured into a glass bottle, it generally cracks because on pouring hot water in the bottle the inner surface heats up and expands more as compared to its outer surface. This unequal expansion between the two surfaces causes a strain and the bottle cracks.
(b). Telephone wires sag in summer because due to heat of the sun, the wire expands and increases in length, thus they sag in summer.
(c). In cold countries water pipes burst in winter because the water has maximum density at 40C and, due to anomalous expansion of water, it expands when the water is cooled to a temperature below 40C
(d). Even when the water in the lakes is frozen, fishes can survive due to anomalous expansion of water, water has maximum density at 40C and this dense water remains at the bottom of the lake and the upper layer of water is less dense and freezes but the temperature of each layer of water below increases by 10C. Thus it is warmer in the lakes below the ice layer. This helps the fishes to survive.
(a). When boiling water is poured into a glass bottle, it generally cracks because on pouring hot water in the bottle the inner surface heats up and expands more as compared to its outer surface. This unequal expansion between the two surfaces causes a strain and the bottle cracks.
(b). Telephone wires sag in summer because due to heat of the sun, the wire expands and increases in length, thus they sag in summer.
(c). In cold countries water pipes burst in winter because the water has maximum density at 40C and, due to anomalous expansion of water, it expands when the water is cooled to a temperature below 40C
(d). Even when the water in the lakes is frozen, fishes can survive due to anomalous expansion of water, water has maximum density at 40C and this dense water remains at the bottom of the lake and the upper layer of water is less dense and freezes but the temperature of each layer of water below increases by 10C. Thus it is warmer in the lakes below the ice layer. This helps the fishes to survive.
Solution 4
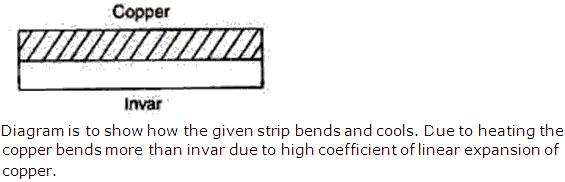
Solution 5
Thermostat is a device for regulating temperature in electric circuits and it is made up of bimetallic strip. Two applications of thermostat are in electric iron and in refrigerators.
Solution 6
When heated, water expands normally but behavior of water is unusual over a small range of temperature between 0 and 40C. This is known as anomalous expansion of water.
Solution 7
Anomalous expansion of water slows down the complete freezing of water in the lake.
Solution 8
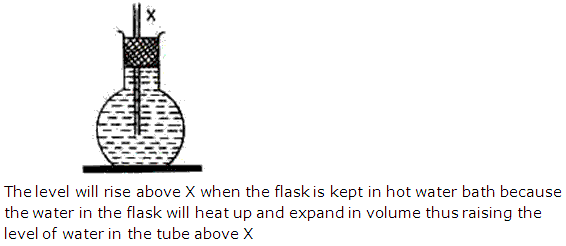
Solution 9
(i). When boiling water is poured into a glass bottle, it generally cracks because on pouring hot water in the bottle the inner surface heats up and expands more as compared to its outer surface. This unequal expansion between the two surfaces causes a strain and the bottle cracks.
(ii). Telephone wires sag in summer because due to heat of the sun, the wire expands and increases in length, thus they sag in summer.
(iii).In cold countries water pipes burst in winter because the water has maximum density at 40C and, due to anomalous expansion of water, it expands when the water is cooled to a temperature below 40C
(iv).Even when the water in the lakes is frozen, fishes can survive due to anomalous expansion of water, water has maximum density at 4(i). When boiling water is poured into a glass bottle, it generally cracks because on pouring hot water in the bottle the inner surface heats up and expands more as compared to its outer surface. This unequal expansion between the two surfaces causes a strain and the bottle cracks.
(ii). Telephone wires sag in summer because due to heat of the sun, the wire expands and increases in length, thus they sag in summer.
(iii).In cold countries water pipes burst in winter because the water has maximum density at 40C and, due to anomalous expansion of water, it expands when the water is cooled to a temperature below 40C
(iv).Even when the water in the lakes is frozen, fishes can survive due to anomalous expansion of water, water has maximum density at 40C and this dense water remains at the bottom of the lake and the upper layer of water is less dense and freezes but the temperature of each layer of water below increases by 10C. Thus it is warmer in the lakes below the ice layer. This helps the fishes to survive.C and this dense water remains at the bottom of the lake and the upper layer of water is less dense and freezes but the temperature of each layer of water below increases by 10C. Thus it is warmer in the lakes below the ice layer. This helps the fishes to survive.
(ii). Telephone wires sag in summer because due to heat of the sun, the wire expands and increases in length, thus they sag in summer.
(iii).In cold countries water pipes burst in winter because the water has maximum density at 40C and, due to anomalous expansion of water, it expands when the water is cooled to a temperature below 40C
(iv).Even when the water in the lakes is frozen, fishes can survive due to anomalous expansion of water, water has maximum density at 4(i). When boiling water is poured into a glass bottle, it generally cracks because on pouring hot water in the bottle the inner surface heats up and expands more as compared to its outer surface. This unequal expansion between the two surfaces causes a strain and the bottle cracks.
(ii). Telephone wires sag in summer because due to heat of the sun, the wire expands and increases in length, thus they sag in summer.
(iii).In cold countries water pipes burst in winter because the water has maximum density at 40C and, due to anomalous expansion of water, it expands when the water is cooled to a temperature below 40C
(iv).Even when the water in the lakes is frozen, fishes can survive due to anomalous expansion of water, water has maximum density at 40C and this dense water remains at the bottom of the lake and the upper layer of water is less dense and freezes but the temperature of each layer of water below increases by 10C. Thus it is warmer in the lakes below the ice layer. This helps the fishes to survive.C and this dense water remains at the bottom of the lake and the upper layer of water is less dense and freezes but the temperature of each layer of water below increases by 10C. Thus it is warmer in the lakes below the ice layer. This helps the fishes to survive.
Solution 10
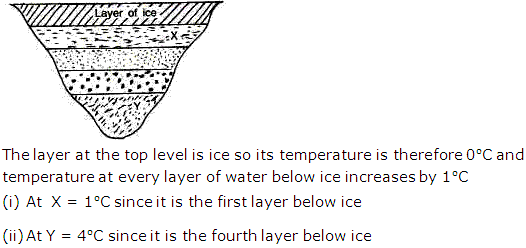
Solution 11
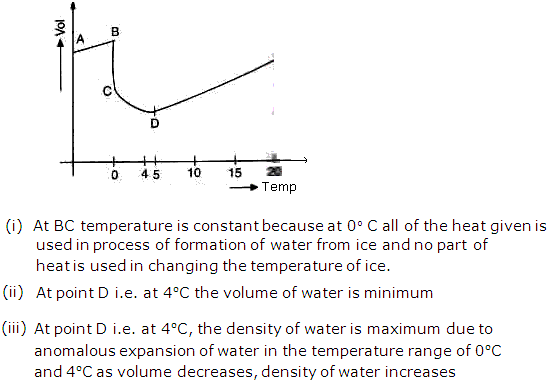
Heat - Thermal Expansion Exercise 203
Solution 12
(i). Maximum
(ii). Increases; decreases
(iii). Coefficient; expansion
(iv). Anomalous expansion of water
(v). Calorie
(iv). Kelvin(K)
(ii). Increases; decreases
(iii). Coefficient; expansion
(iv). Anomalous expansion of water
(v). Calorie
(iv). Kelvin(K)
Solution 13
We should heat the neck of the bottle because due to heating the neck will expand and loosen the stopper stuck in the neck. In this way, we can easily remove the stopper.
Solution 14
When a liquid is heated in a flask. Due to heat, the flask expands along with the liquid, thus providing more space for the liquid. Hence the level of the liquid falls. Thus this dropped level shows the apparent expansion of the liquid. The actual expansion of the liquid, when heated, is the real expansion.
Solution 15
No, the both liquids i.e. mercury and alcohol will not have same volume on heating as they have different coefficient of expansions, thus they expand differently on heating at same temperature range. Alcohol expands more than mercury.
Solution 16
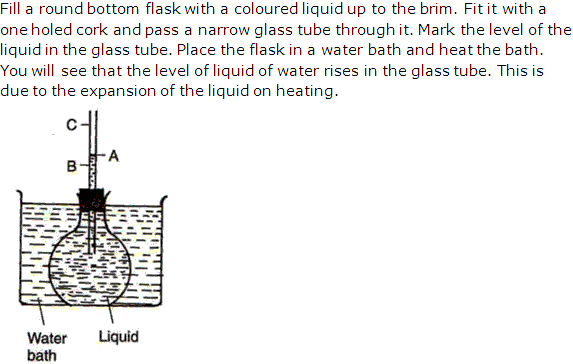
Solution 17
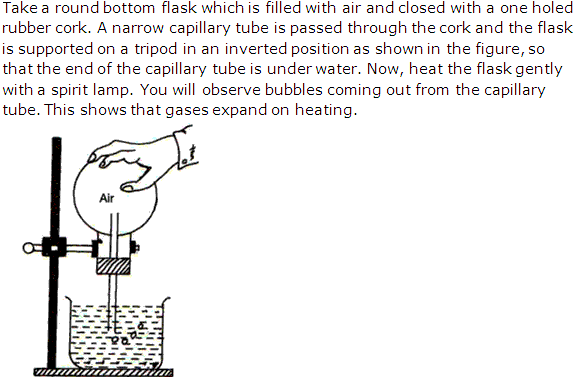
Solution 18
A hot glass chimney often crack when a drop of water falls on it because the glass of the chimney expands due to constant heating by the flame of the chimney but as a cooler water drop falls on the glass, the outer surface contracts more than the inner side of the glass. . This unequal contraction between the two surfaces causes a strain and the glass cracks.
Solution 19
No, we cannot fuse iron wire in glass rod because glass is bad conductor of heat and it does not allow heat to pass through it and iron wire would not be able to get sufficient heat to fuse.
Solution 20
We should heat the neck of the bottle because due to heating the neck will expand and loosen the stopper stuck in the neck. In this way, we can easily remove the stopper from the bottle.
Solution 21
Two substances that expand on heating are water and iron.
Solution 22
Two substances that contract on heating are plastic and poly-ethene.
Solution 23
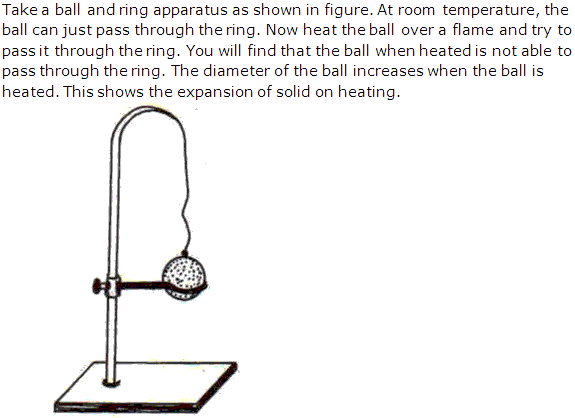
Solution 24
The joints in metal pipes loosen in summers because the joints get more space to expand due to the heat in summers as compared to the pipes therefore the joints become loose in summers
Solution 25
The increase in length of a rod depends on the following factors
(a). material of the rod
(b). Original length of the rod
(c). Rise in temperature
(a). material of the rod
(b). Original length of the rod
(c). Rise in temperature
Solution 26
The given statement states that the change in length of the metal will be 0.000016 m when its temperature changes by 10C.
Solution 27
On heating the copper washer
(a). Its internal diameter will increase as the copper washer expands outwards
(b). Volume will increase as the size of the metal expands on heating
(c). Mass will remain the same as the mass does not change with change in temperature
(d). Density will decrease as the volume of the washer increases on heating and density is inversely proportional to volume
(e). External diameter will increase because the copper washer expands outwards
(a). Its internal diameter will increase as the copper washer expands outwards
(b). Volume will increase as the size of the metal expands on heating
(c). Mass will remain the same as the mass does not change with change in temperature
(d). Density will decrease as the volume of the washer increases on heating and density is inversely proportional to volume
(e). External diameter will increase because the copper washer expands outwards
Solution 28
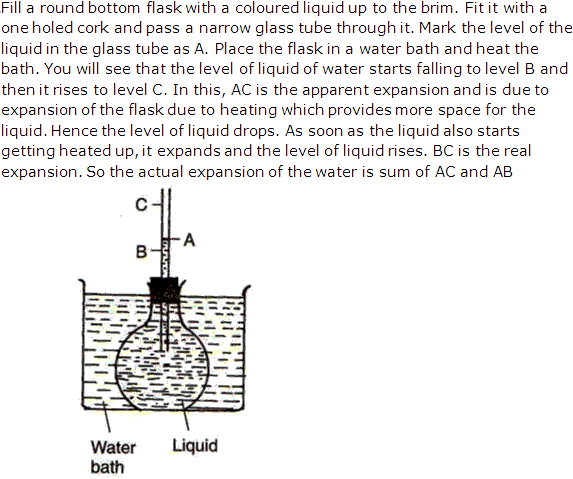
Solution 29
At 40C, water has the maximum density due to anomalous expansion.
Solution 30

