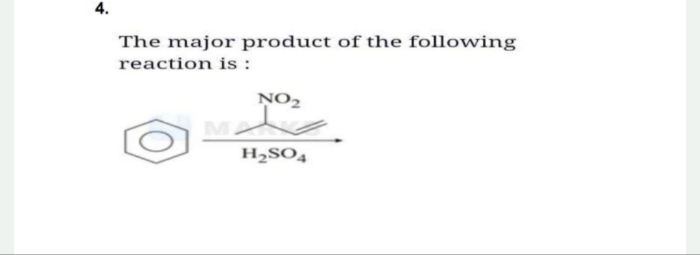
NEET Class neet Answered
Why this compound is not antiaromatic while still having 4π electrons and continuous chain resonance
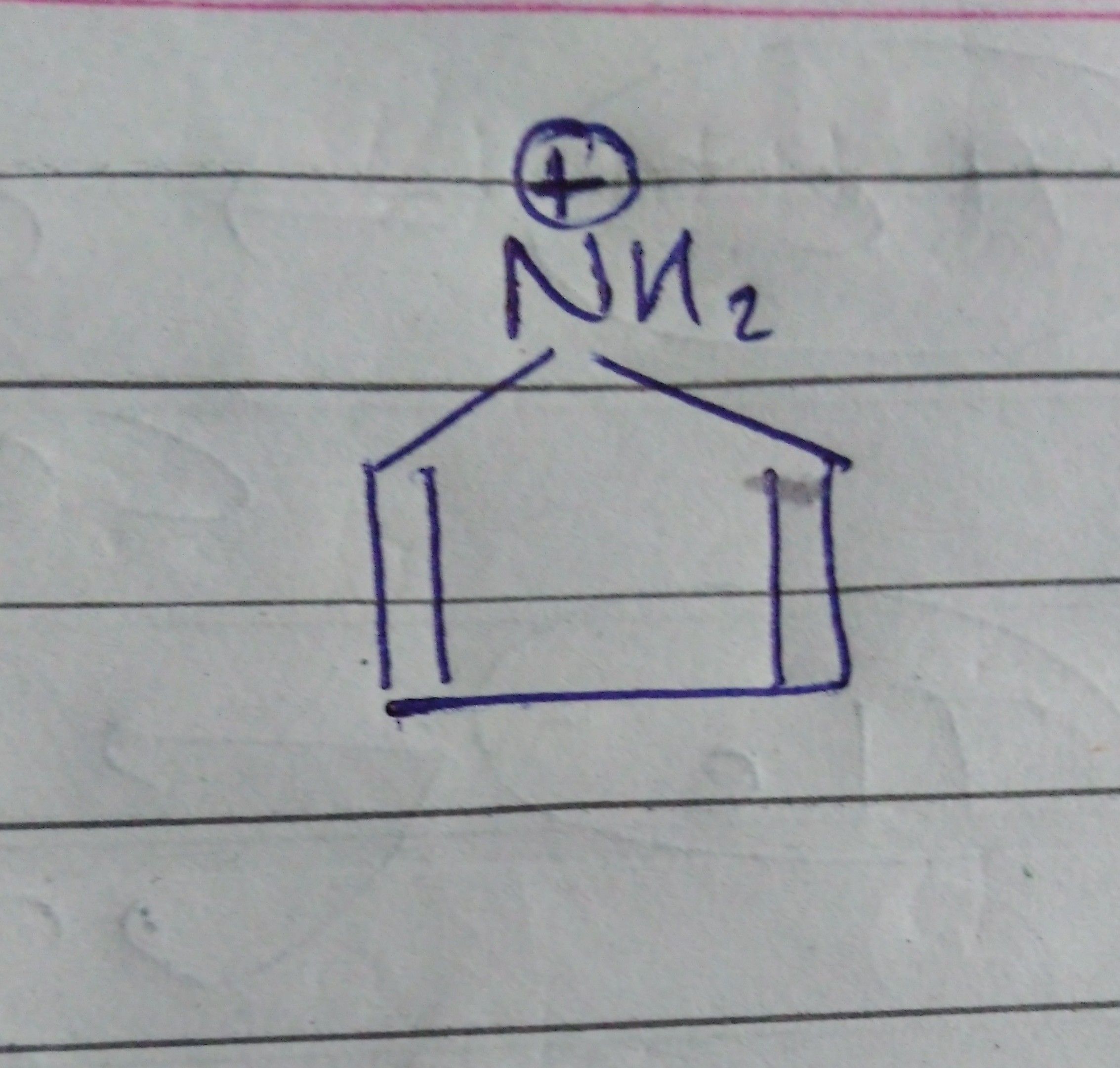
Asked by musira29rahman | 18 Aug, 2019, 04:40: PM

Pyrrole is a 5 membered heterocycle which is also aromatic.

The protonated pyrrole would no longer be aromatic, because there would no longer be 6∏ electrons, and also the N would have to be sp3 (4 bonds) and so have no p orbital for the required ring of p orbitals for aromaticity.
Answered by Ramandeep | 20 Aug, 2019, 01:07: PM
Concept Videos
NEET neet - Chemistry
Asked by ghousiakaneez | 03 Apr, 2024, 12:55: PM
NEET neet - Chemistry
Asked by paritoshanjare23 | 26 Jan, 2024, 09:52: PM
NEET neet - Chemistry
Asked by anmol180018 | 13 Jan, 2024, 10:39: PM
NEET neet - Chemistry
Asked by somudharani93 | 13 Jan, 2024, 09:45: PM
NEET neet - Chemistry
Asked by ayushsa796 | 23 Oct, 2023, 09:03: PM
NEET neet - Chemistry
Asked by redefinebiology | 11 Mar, 2023, 11:21: PM
NEET neet - Chemistry
Asked by shathwala844 | 27 Jun, 2022, 10:12: AM
NEET neet - Chemistry
Asked by mdaffanmallick28 | 26 May, 2022, 09:52: PM
NEET neet - Chemistry
Asked by mdaffanmallick28 | 26 May, 2022, 09:51: PM
NEET neet - Chemistry
Asked by pallavikumarimddb | 06 May, 2022, 01:54: AM