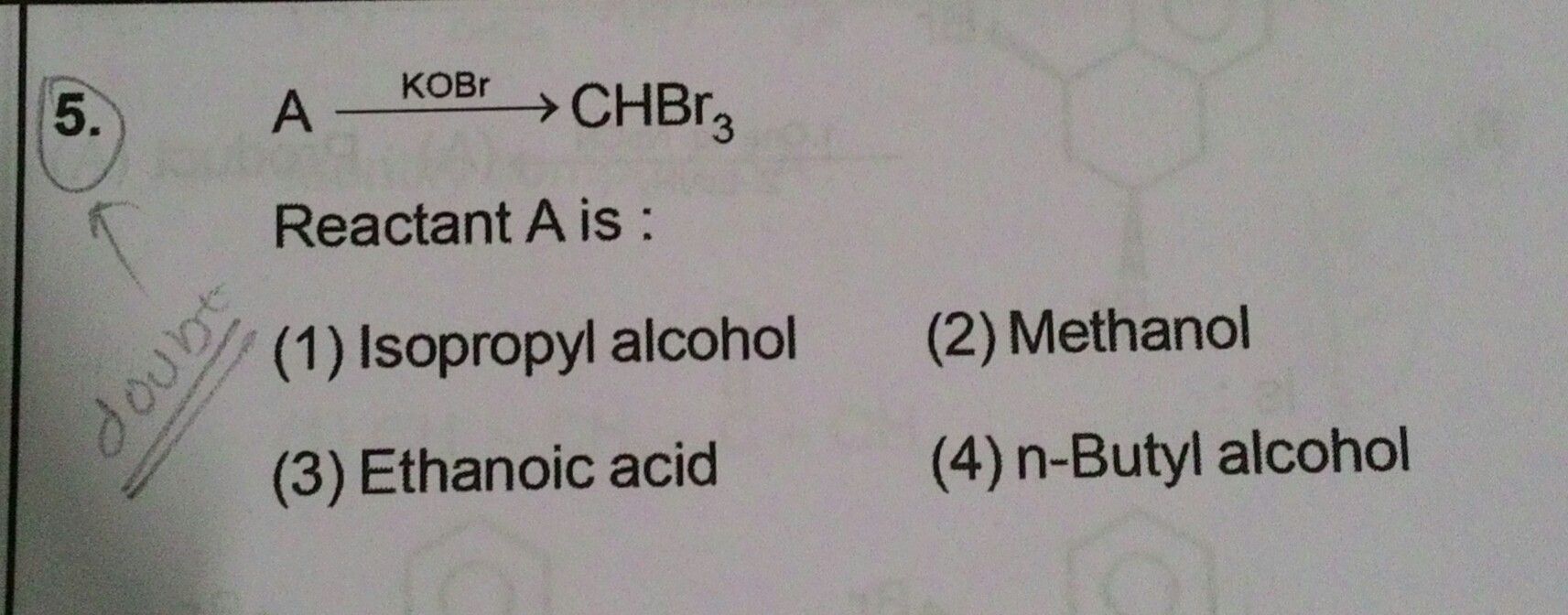NEET Class neet Answered
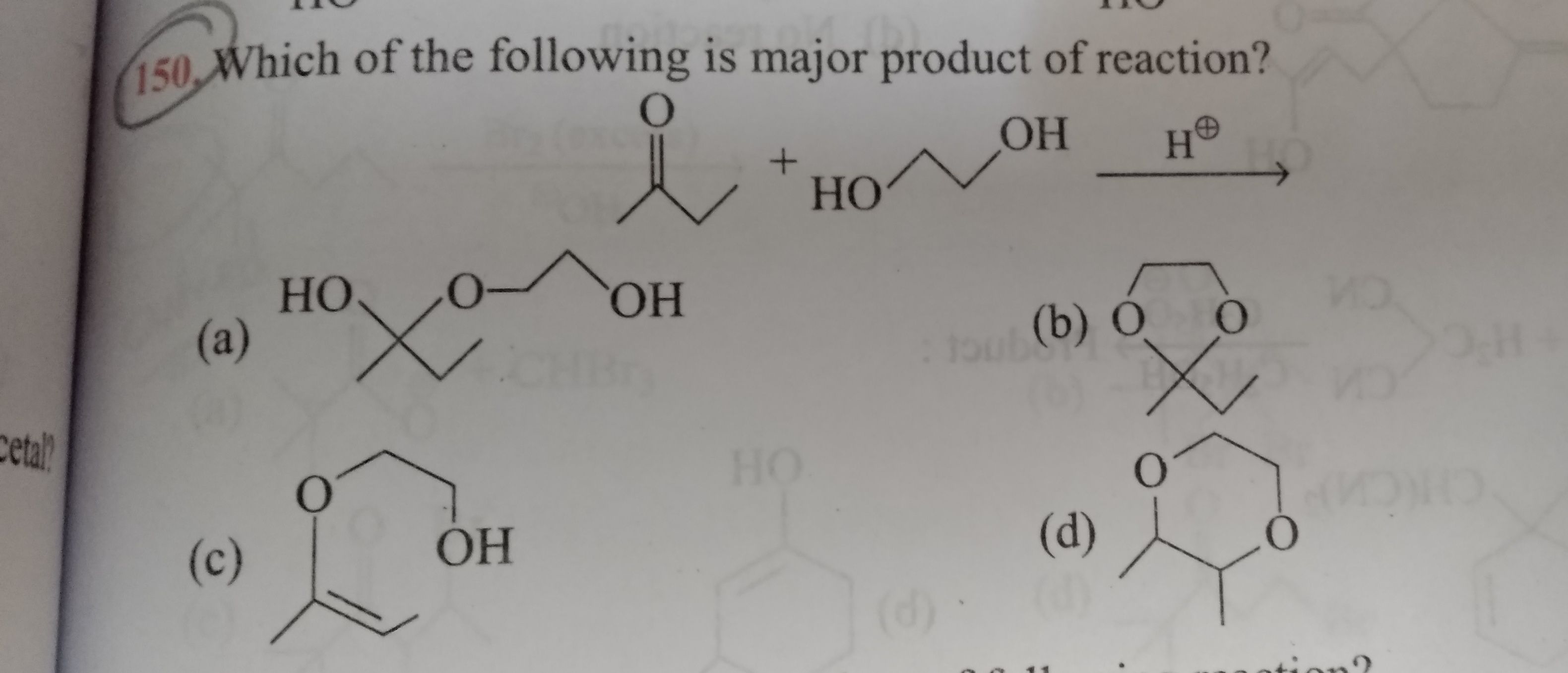
The strongest acid among the following compounds is
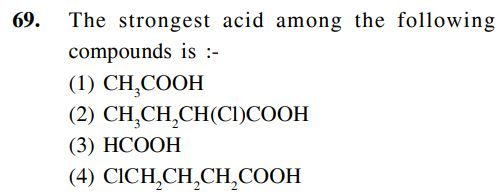
Asked by Mrinal | 14 May, 2019, 09:23: AM
An electron withdrawing group is increases the strength of acid by attracting the electron density towards itself. Due to this the the -O-H bond becomes weak resulting release of H+ ion.
while +I effect increases the electron density and hence decreases the strength of acid.
As well as the -I effect is decreases as distane is increases.
Hence the strongest acid among the given compounds is
option B
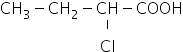
Answered by Ramandeep | 14 May, 2019, 10:42: AM
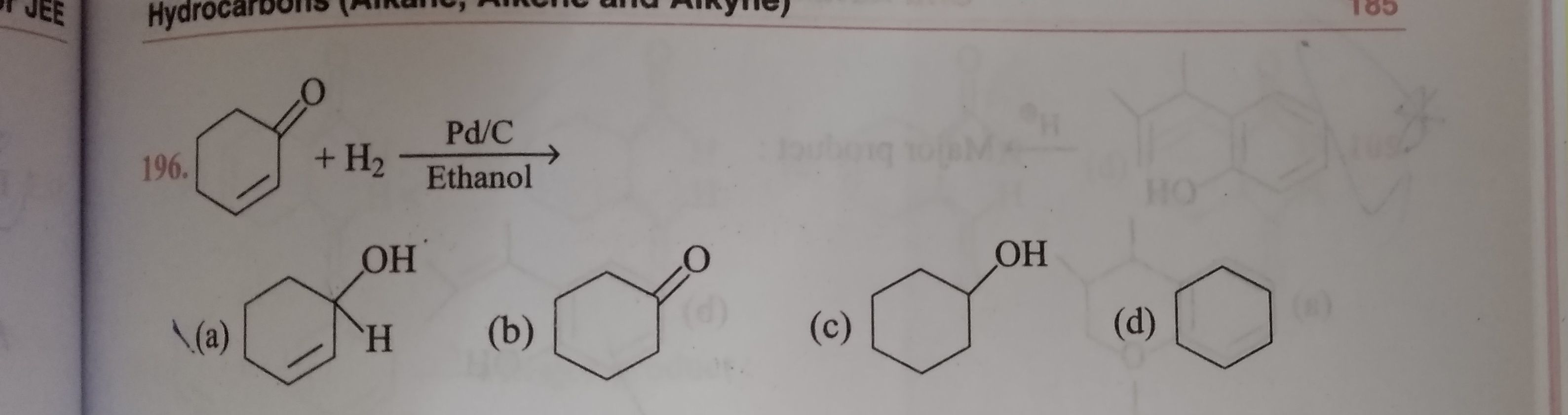
NEET neet - Chemistry
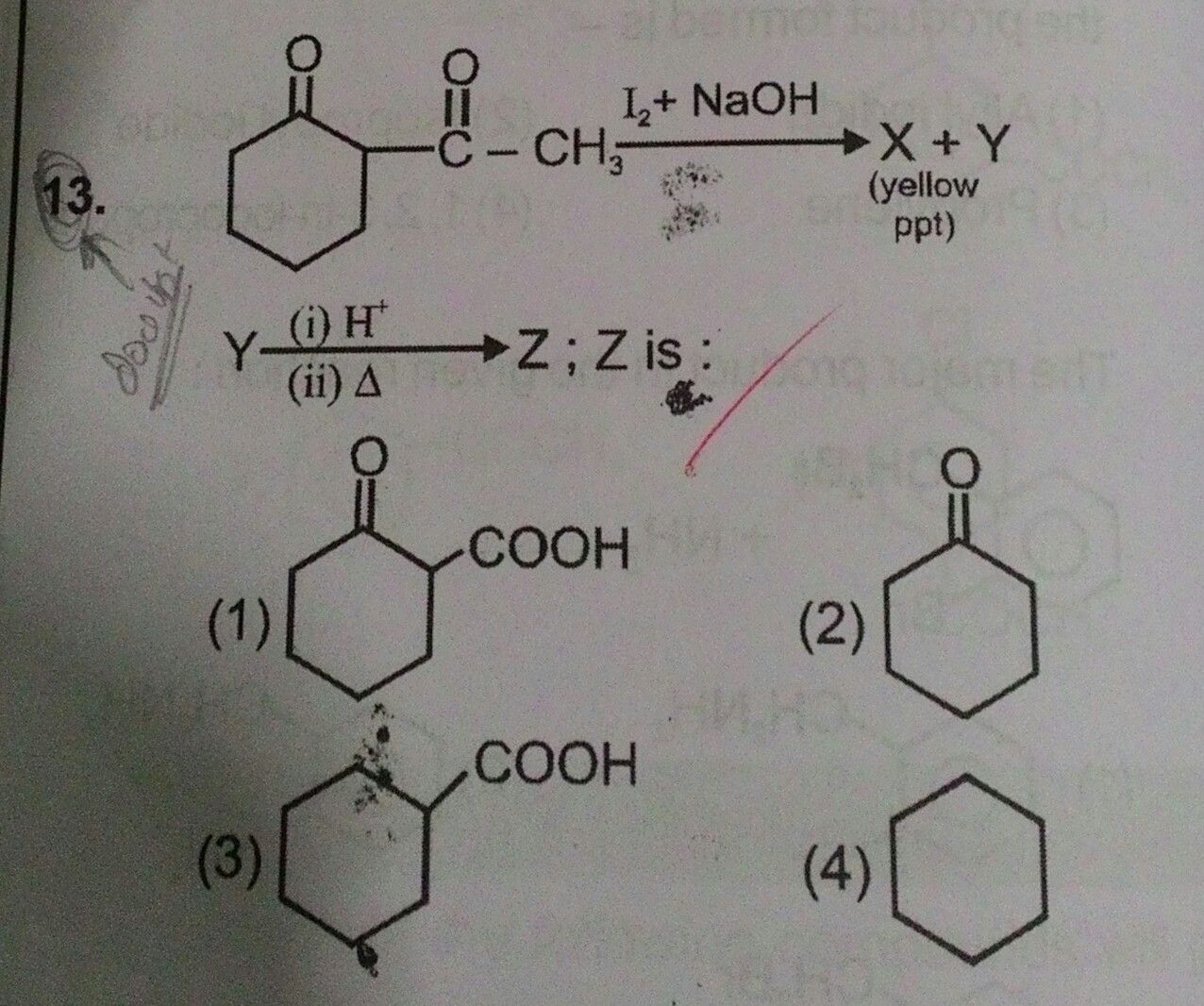
Asked by prakriti12oct | 01 Sep, 2020, 12:09: AM
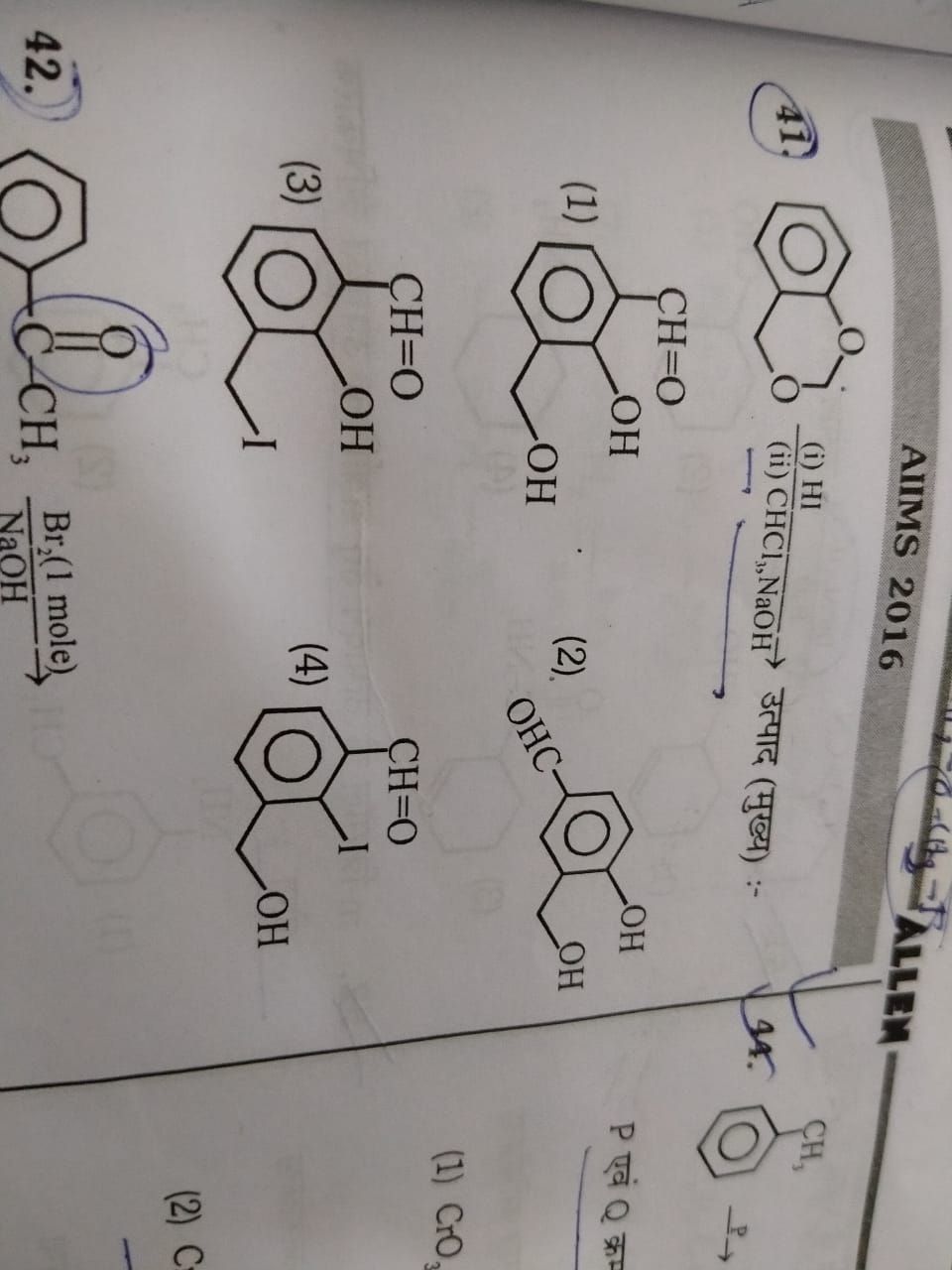
NEET neet - Chemistry
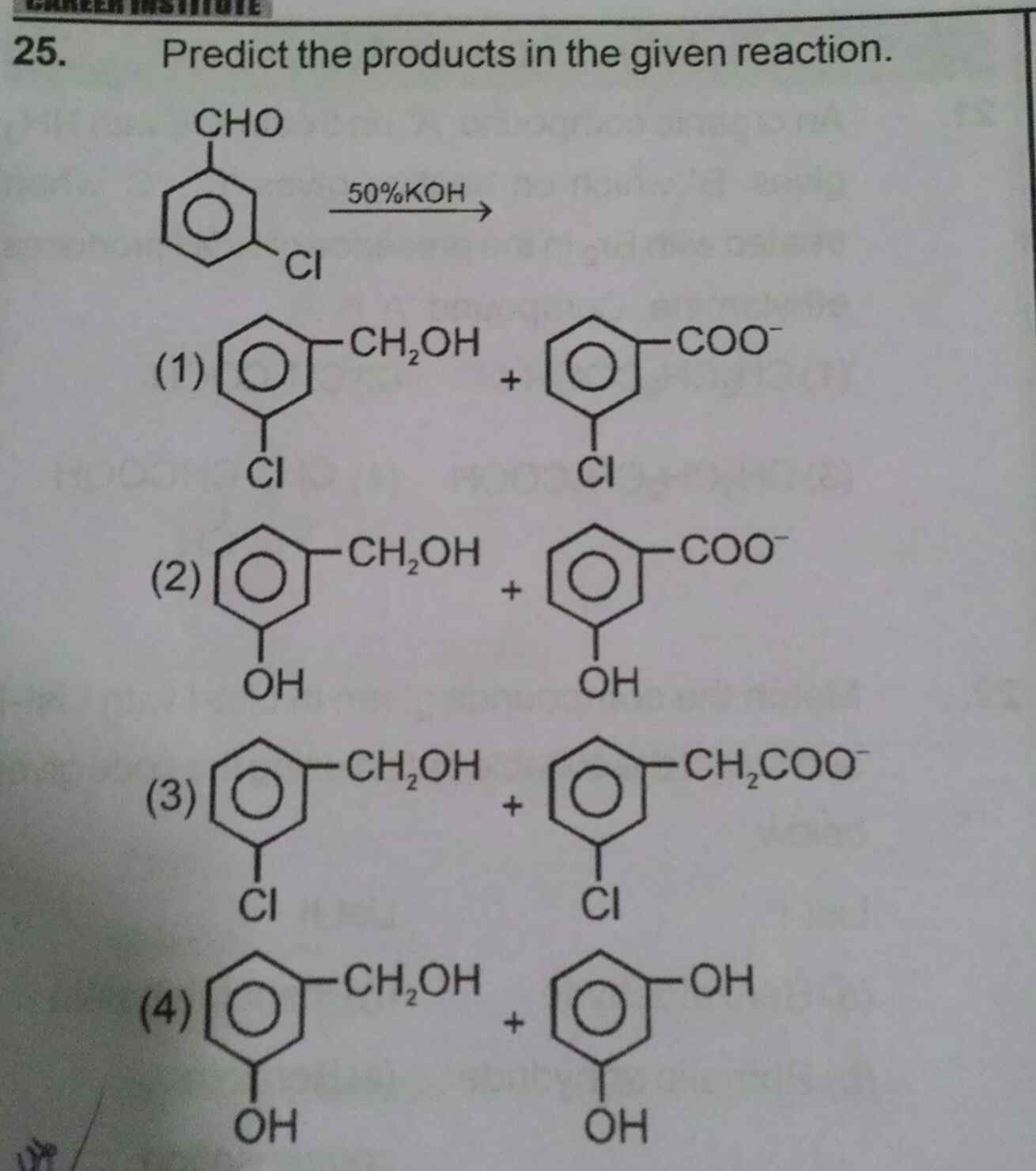
Asked by subhrojyotighosh8 | 22 Jul, 2020, 11:24: PM
NEET neet - Chemistry
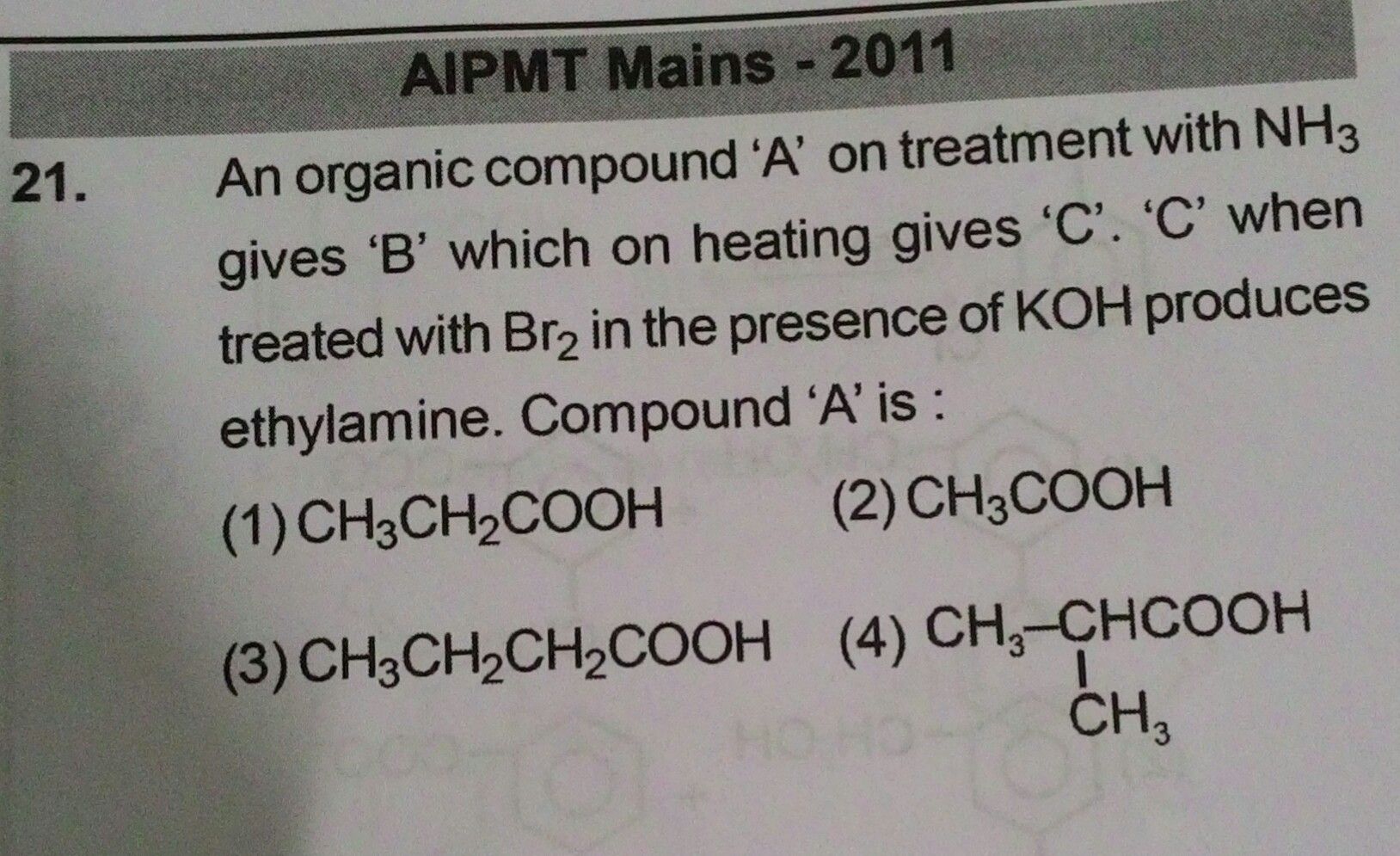
Asked by subhrojyotighosh8 | 12 Jul, 2020, 10:40: PM
NEET neet - Chemistry
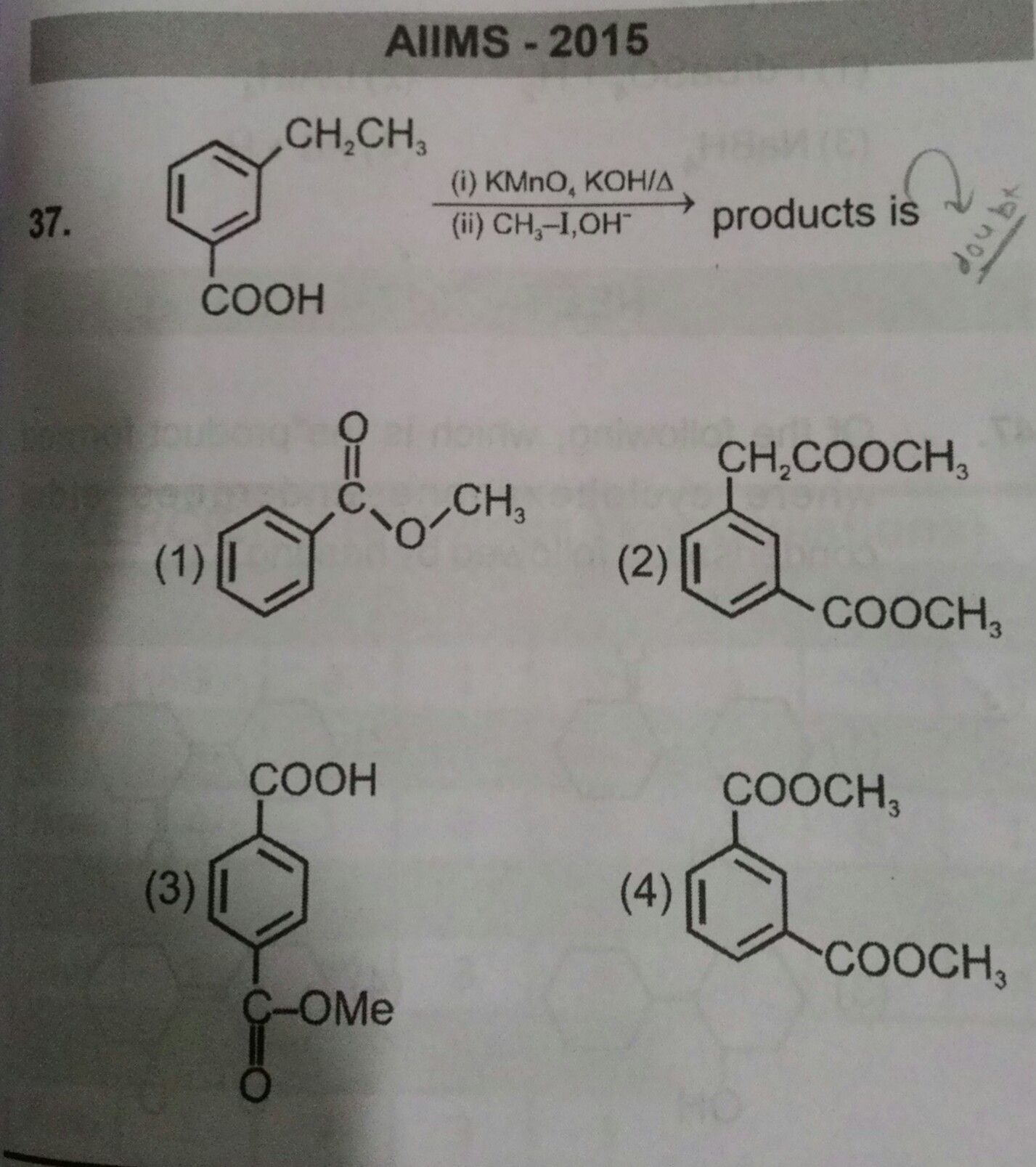
Asked by Prashant DIGHE | 27 Feb, 2020, 09:45: PM
NEET neet - Chemistry
Asked by Prashant DIGHE | 24 Feb, 2020, 10:02: PM
NEET neet - Chemistry
Asked by Prashant DIGHE | 23 Feb, 2020, 10:26: PM
NEET neet - Chemistry
Asked by Prashant DIGHE | 20 Feb, 2020, 10:16: PM
NEET neet - Chemistry
Asked by Prashant DIGHE | 19 Feb, 2020, 10:46: PM
NEET neet - Chemistry
Asked by Prashant DIGHE | 19 Feb, 2020, 10:28: PM