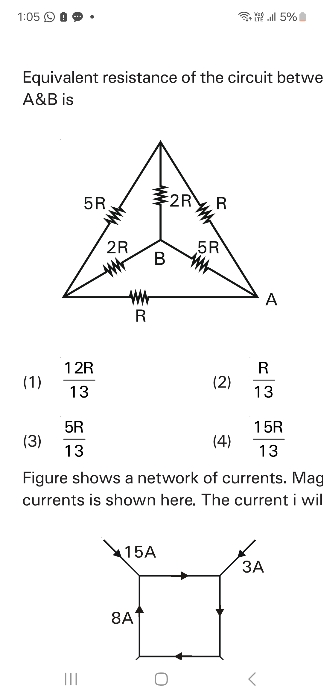
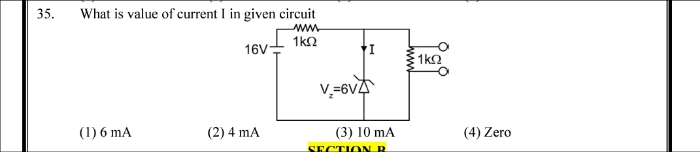
NEET Class neet Answered
⭕Refer to attachment ⭕
Explain me..why we can't use formula directly as
W = -2.303PV.log(P1/P2)

Asked by jhajuhi19 | 03 Mar, 2020, 07:54: AM
Yes the formula can be used
W = 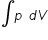
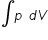
if p V = n R T ..............(1)
where p is pressure , V is volume , n is number of moles, R is gas constant and T is absolute temperature.
differentiating eqn.(1), p dV + V dp = 0 or p dV = - V dp = -(nRT) ( dp/p)
Hence workone W =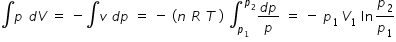
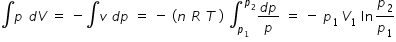
Hence workdone , W = - 2 × 1.013 × 105 ×5 × 10-3 ln( 1/2) = 702 J
( SI units are used, pressure in Pa and volume in m3 )
Answered by Thiyagarajan K | 03 Mar, 2020, 08:37: AM
Application Videos
NEET neet - Physics
Asked by bidyutpravarout79 | 26 Apr, 2024, 09:40: PM
NEET neet - Physics
Asked by ramanjaneyuluoguru | 25 Apr, 2024, 04:18: PM
NEET neet - Physics
Asked by shatakshibhatt9 | 20 Apr, 2024, 07:52: PM
NEET neet - Physics
Asked by praveenpriya000079 | 18 Apr, 2024, 07:24: AM
NEET neet - Physics
Asked by gouranshi84 | 17 Apr, 2024, 05:23: PM
NEET neet - Physics
Asked by sojusvi | 17 Apr, 2024, 01:12: PM