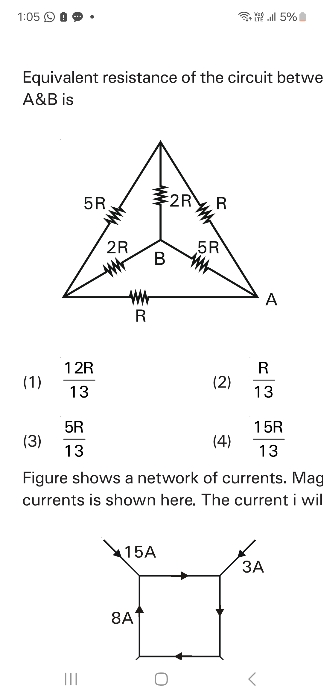
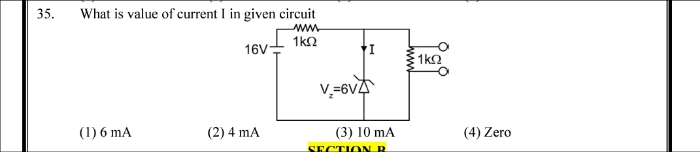
NEET Class neet Answered
⭕Answer this question (62) with Explaination...Thanks🙂
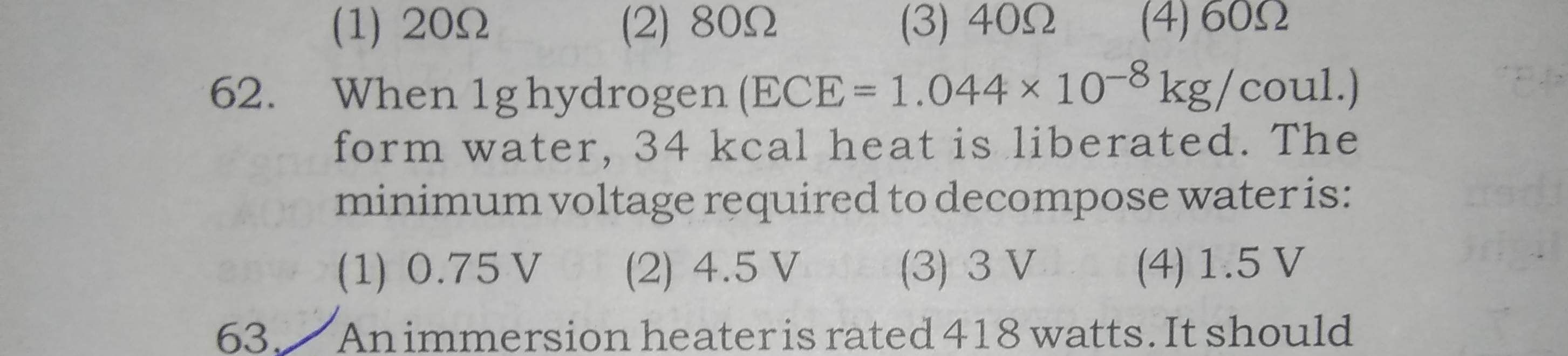
Asked by jhajuhi19 | 07 Apr, 2020, 04:44: AM
We are given that electro chemical equivalent of electrolysis of water is 1.044 × 10-8 kg/C
Hence if 1.044 × 10-8 kg of water is undergoing electrolysis, quantity of charge created is 1 C
if 1 g of water is undergoing electrolysis, then quantity of charge created is [ 10-3 / ( 1.044 × 10-8 ) ] = 9.58 × 104 C
Energy released in electrolysis of 1 g hydrogen = 34 kcal = 34 × 103 × 4.2 J = 142.8 × 103 J
Hence 142.8 × 103 J energy is required to pass 9.58 × 104 C charge across electordes.
Potential difference between electrode is workdone to move unit charge across electrodes.
Hence potetial difference between electrodes = ( 142.8 × 103 / 9.58 × 104 ) J/C ≈ 1.5 V
Answered by Thiyagarajan K | 07 Apr, 2020, 09:53: AM
Application Videos
NEET neet - Physics
Asked by bidyutpravarout79 | 26 Apr, 2024, 09:40: PM
NEET neet - Physics
Asked by ramanjaneyuluoguru | 25 Apr, 2024, 04:18: PM
NEET neet - Physics
Asked by shatakshibhatt9 | 20 Apr, 2024, 07:52: PM
NEET neet - Physics
Asked by praveenpriya000079 | 18 Apr, 2024, 07:24: AM
NEET neet - Physics
Asked by gouranshi84 | 17 Apr, 2024, 05:23: PM
NEET neet - Physics
Asked by sojusvi | 17 Apr, 2024, 01:12: PM