JEE Class main Answered
Where H+ Attack How To Know
Asked by sahusohan179 | 12 Jan, 2023, 05:24: AM
Dear Student,
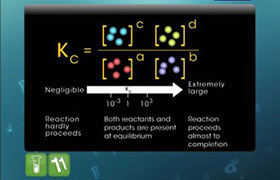
H+ will attack the molecule at the position whiere electron density is more.
It attackes in such a way that the resulting structure would be more stable.
More the resonance, more the stability.
For example,
Answered by | 12 Jan, 2023, 12:37: PM
Application Videos
Concept Videos
JEE main - Chemistry
Asked by neerajavuppala1983 | 23 Jul, 2024, 22:49: PM
JEE main - Chemistry
Asked by tanniruv133 | 03 Jul, 2024, 18:50: PM
JEE main - Chemistry
Asked by heyyyyy | 12 Jun, 2024, 19:18: PM
JEE main - Chemistry
Asked by hv5594265 | 12 Jun, 2024, 11:59: AM
JEE main - Chemistry
Asked by rupalibhange1987 | 11 Jun, 2024, 20:00: PM
JEE main - Chemistry
Asked by gajju8493 | 11 Jun, 2024, 15:09: PM
JEE main - Chemistry
Asked by chakrabortymithu041 | 29 May, 2024, 18:45: PM