CBSE Class 10 Answered
Sulphur dioxide is covalent compound then how it can form ions when dissolved in water?
Asked by govtsecschoolnayaganv051 | 27 Feb, 2018, 10:49: PM
Don't get confused in covalent compounds and ionic compounds,
before coming to the answer first, understand bond type in SO2 molecule.
SO2 has covalent bonding but because of electronegativity difference between S and O, the covalent bond becomes polar.
Electronegativity of O is 3.5 and sulphur has 2.6, the difference is 0.9 (3.5 - 2.6 = 0.9).
So, the bond is somewhat polar but still covalent.
Hence because of polar bonding, it dissolved in polar solvents like water and form ions.
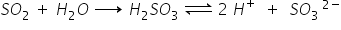
Answered by Ramandeep | 28 Feb, 2018, 10:13: AM
Application Videos
Concept Videos
CBSE 10 - Chemistry
Asked by aamjdkhan640 | 03 Feb, 2022, 04:54: PM
CBSE 10 - Chemistry
Asked by anushka.boraste | 24 Jan, 2022, 08:12: AM
CBSE 10 - Chemistry
Asked by ishusain2005 | 21 Aug, 2021, 07:19: AM
CBSE 10 - Chemistry
Asked by sudamamaheshwari5 | 28 Jun, 2021, 06:05: AM
CBSE 10 - Chemistry
Asked by diyavinodj | 19 Feb, 2021, 09:06: PM
CBSE 10 - Chemistry
Asked by singhakansha496 | 21 Dec, 2019, 03:39: PM
CBSE 10 - Chemistry
Asked by Nash9837.a | 25 Sep, 2019, 06:15: AM
CBSE 10 - Chemistry
Asked by saradamahapatra9999 | 09 Jul, 2019, 10:09: AM
CBSE 10 - Chemistry
Asked by ashishaman25082004 | 21 Jun, 2019, 07:40: PM
CBSE 10 - Chemistry
Asked by prasutally | 24 May, 2019, 01:44: PM





