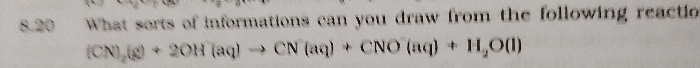NEET Class neet Answered
In steam distillation, why the flask containing distillate is also heated (under bunsen burner) if evaporation is already done by passing steam over it ??
Asked by myindiaisbad | 04 Mar, 2023, 12:02: PM
Dear student,
In steam distllation, the distillate contains both organic liquid to be separated along with water vapours.
This mixture has to be kept at boiling.
Please note that, here only organic compound is not getting heated hence it will not get decomposed.
Water vapours along with vapours of volatile compound come out and the volatile part is separated later.
The water vapours should not get condensed back to liquid water.
Hence, there is a need to heat the flask of distillate to be heated at low temperature.
Answered by | 04 Mar, 2023, 15:22: PM
Application Videos
Concept Videos
NEET neet - Chemistry
Asked by narayandhareppanavar753 | 03 Jul, 2024, 20:03: PM
NEET neet - Chemistry
Asked by khanhazoor446 | 07 Jun, 2024, 12:52: PM
NEET neet - Chemistry
Asked by kishusambhar | 05 Jun, 2024, 11:31: AM
NEET neet - Chemistry
Asked by princejewel712 | 28 May, 2024, 08:26: AM
NEET neet - Chemistry
Asked by priyankasahoo0086 | 22 May, 2024, 06:36: AM
NEET neet - Chemistry
Asked by kanishksingh538 | 22 May, 2024, 00:27: AM
NEET neet - Chemistry
Asked by ruchisharmatbn | 20 May, 2024, 17:53: PM
NEET neet - Chemistry
Asked by kanika4070 | 17 May, 2024, 21:45: PM
NEET neet - Chemistry
Asked by sahumansi | 12 May, 2024, 09:37: AM