ICSE Class 10 Answered
Give reason why is roasting generally carried out on sulfuric ores and not on carbonate ores
Asked by sardagaura | 10 Jun, 2019, 12:17: PM
t is easier to obtain metals from their oxides (by reduction) than from carbonates or sulphides.
So before reduction can be done, the ore is converted into metal oxide.
The concentrated ores can be converted into metal oxide by calcination or roasting.
Calcination: It is the process in which a carbonate ore is heated strongly in the absence of air toconvert it into a metal oxide.
Example:When zinc carbonate is heated strongly in the absence of air, it decomposes to form zinc oxideand carbon dioxide
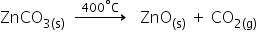
Roasting: It is the process in which a sulphide ore is strongly heated in the presence of air to convert it into a metal oxide.
This is done in case of sulphide ores so as to remove sulphur in the form of SO2 and to obtained corresponding metal oxide.
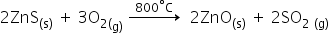
Answered by Ravi | 10 Jun, 2019, 01:05: PM
Application Videos
Concept Videos
ICSE 10 - Chemistry
Asked by sardagaura | 10 Jun, 2019, 12:17: PM
ICSE 10 - Chemistry
Asked by pb_ckt | 20 Mar, 2019, 01:29: PM




