ICSE Class 9 Answered
give balanced molecular eqution for : 1) .one reactant gives 2 product 2). one reactant gives 3 products 3). two reactant gives 2 product 4.) two reactant gives 4 products
Asked by garimaydv2005 | 15 Sep, 2020, 16:10: PM
Balanced chemical equations for these four cases-
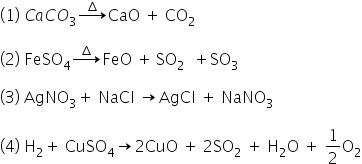
Answered by Ravi | 15 Sep, 2020, 22:57: PM
Application Videos
Concept Videos
ICSE 9 - Chemistry
Asked by 2016159 | 06 Jul, 2024, 12:45: PM
ICSE 9 - Chemistry
Asked by siddheshmathur | 12 Jul, 2022, 01:14: AM
ICSE 9 - Chemistry
Asked by kavisrilalu | 06 Nov, 2021, 13:49: PM
ICSE 9 - Chemistry
Asked by jailakshmitradersdnn | 04 Mar, 2021, 00:00: AM
ICSE 9 - Chemistry
Asked by garimaydv2005 | 15 Sep, 2020, 16:10: PM
ICSE 9 - Chemistry
Asked by swarnatottaramudi | 21 Aug, 2020, 10:34: AM
ICSE 9 - Chemistry
Asked by arpanchavan47.9spicertl | 17 Jun, 2020, 17:19: PM
ICSE 9 - Chemistry
Asked by bhoomikapoor200 | 16 May, 2020, 22:31: PM
ICSE 9 - Chemistry
Asked by sayantikasingh3318.9sdatl | 15 May, 2020, 20:32: PM










