ICSE Class 8 Answered
Catalyst and their actions
A catalyst is a substance which either increases or decreases the rate of a chemical reaction without itself undergoing any chemical change during the reaction.
Some chemical reactions need a catalyst to change the rate of the reaction.
1) Positive catalyst: It increases the rate of reaction.
Examples:
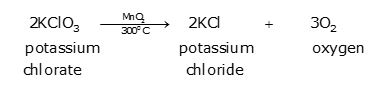
On heating to 700°C, potassium chlorate decomposes to evolve oxygen gas.
But when manganese dioxide is mixed with it, the decomposition takes place at a much lower temperature, at about 300°C. In this reaction, manganese dioxide acts as a catalyst and remains unaffected.
2) Negative catalyst: It decreases the rate of reaction.
Example:
Phosphoric acid acts as a negative catalyst to decrease the rate of decomposition of hydrogen peroxide.
Alcohols also act as a negative catalyst in certain chemical reactions.