ICSE Class 10 Answered
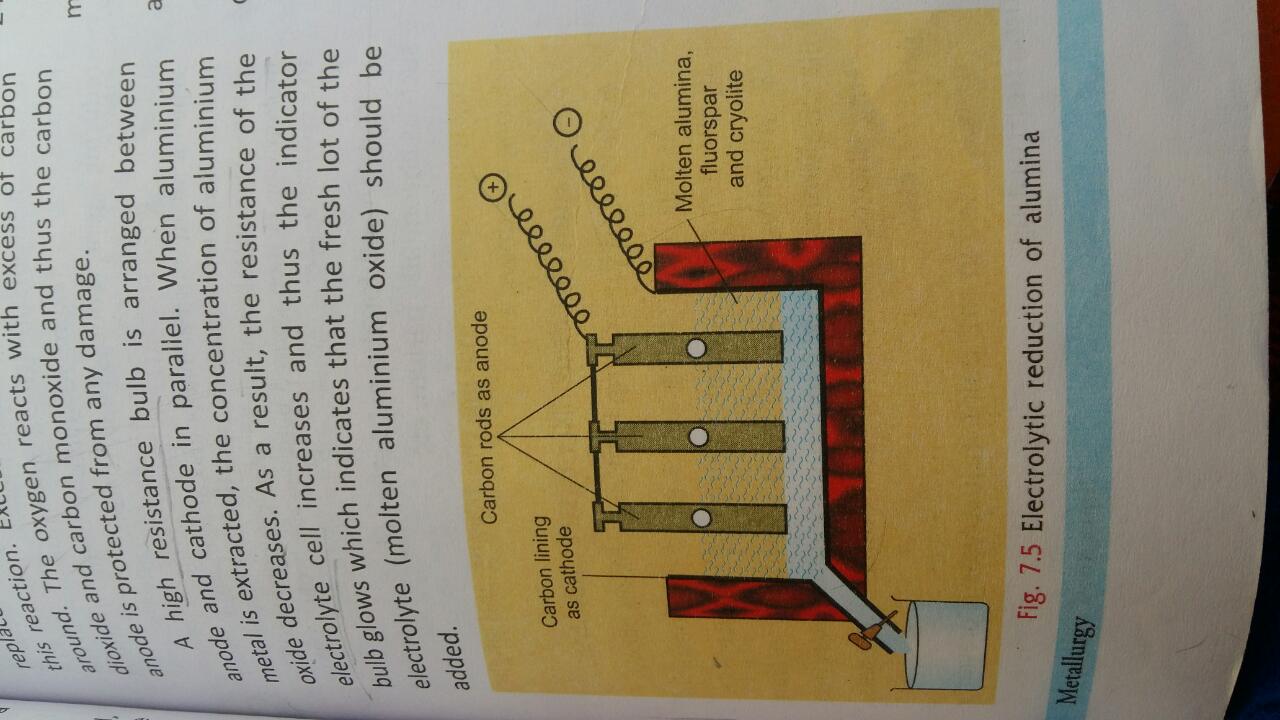
(a) Aluminium is extracted from its chief ore, bauxite. The ore is first purified and then the metal is extracted from it by electrolytic reduction.(i) Write three balanced chemical equations for the purification of bauxite by Hall's process. (ii) Name a chemical used for dissolving aluminium oxide. In which state of sub-divison is the chemical used? (iii) Write an equation for the reaction which takes place at the anode during the extraction of aluminium by the electrolysis process. (b) From the list of characteristics given below, select the five which are relevant to non-metals and their compounds:A. DuctileB. Conduct electricityC. BrittleD. Acidic oxidesE. basic oxidesF. Discharged at anodeG. Discharged at cathodeH. Ionic chloridesI. Covalent chloridesJ. Reaction with dilute sulphuric acid yields hydrogenK. 1, 2 or 3 valence electronsL. 5, 6 or 7 valence electrons (Write the five letters corresponding to the correct characteristics)
Asked by Topperlearning User | 12 May, 2016, 04:44: PM

Answered by | 12 May, 2016, 06:44: PM
Application Videos
Concept Videos
ICSE 10 - Chemistry
Asked by jamsandekar.leena | 17 Mar, 2019, 07:12: PM
ICSE 10 - Chemistry
Asked by Tech guides by | 29 Jan, 2019, 06:35: PM
ICSE 10 - Chemistry
Asked by ruthikreddy2004 | 06 Dec, 2018, 03:55: PM
ICSE 10 - Chemistry
Asked by 21janhvi.verma | 24 Sep, 2018, 12:32: PM
ICSE 10 - Chemistry
Asked by Prvati | 26 Jan, 2018, 07:18: AM
ICSE 10 - Chemistry
Asked by lovemaan5500 | 06 Jan, 2018, 03:11: PM