Class 10 LAKHMIR SINGH AND MANJIT KAUR Solutions Chemistry Chapter 3 - Metals and Non-metals
Metals and Non-metals Exercise 131
Solution 1
Metal - Mercury.
Non metal - Bromine.
Solution 2
Metals are electropositive elements because they can form positive ions by losing electrons.
Non-metals are electronegative elements because they can form negative ions by gaining electrons.
Solution 3
(a) Aluminium.
(b) Oxygen.
Metals and Non-metals Exercise 132
Solution 4
Cesium.
Solution 5
Lead.
Solution 6
False.
Solution 7
(i) Aluminium.
(ii) Copper.
Solution 8
Iron nail gets covered with a red-brown coating of copper metal; The blue colour of copper sulphate solution fades gradually.
Solution 9
Aqua-regia is a freshly prepared mixture of one part of concentrated nitric acid and 3 parts of concentrated hydrochloric acid. Gold and platinum dissolve in aqua-regia
Solution 10
(a) Carbon dioxide and sulphur dioxide.
(b) Sodium oxide and magnesium oxide.
Solution 11
Amphoteric oxides.
Solution 12
Aluminium and zinc.
Solution 13
Copper coin will get a shining greyish white coating of silver metal. The color of the solution will turn blue.
Solution 14
(a) High thermal conductivity.
(b) High electrical conductivity.
Solution 15
Sodium hydride, NaH
Hydrogen sulphide, H2S
Solution 16
(a) Gold.
(b) Potassium.
(c) Mercury.
Solution 17
Copper.
Solution 18
(a) Sodium.
(b) White phosphorus.
Solution 19
(a)
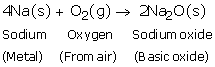
(b)
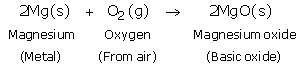
Solution 20
(a) Aluminium and copper.
(b) Copper and aluminium.
(c) Gold and silver.
Solution 21
Aluminium foil.
Solution 22
(a) Hydrogen.
(b) Hydrogen.
(c) Carbon.
(d) Nitrogen.
(e) Sulphur.
Solution 23
(a) Metals are malleable.
(b) Non-metals are non-malleable.
Solution 24
Brittleness is the property of being brittle i.e. breaking easily.
Non-metals show brittleness.
Solution 25
When a strip of zinc metal is put in copper sulphate solution, then the blue colour of copper sulphate solution fades gradually and red brown coating of copper is deposited on zinc strip.
Solution 26
When a strip of copper metal is immersed in silver nitrate solution, the solution gradually becomes blue and a shining greyish-white deposit of silver metal is formed on copper strip.
Solution 27
When iron nails are placed in copper sulphate solution, the blue colour of copper sulphate solution fades gradually and red-brown copper metal is formed.
Solution 28
If a strip of silver metal is kept immersed in copper sulphate solution for some time, then no reaction occurs. This shows that silver is not able to displace copper from copper sulphate solution.
Solution 29
Blue color of copper sulphate is destroyed because iron displaces copper from copper sulphate solution as iron is more reactive than copper.
Metals and Non-metals Exercise 133
Solution 30
Diamond.
Solution 31
Since graphite is a good conductor of electricity, it is used in making electrodes.
Solution 32
Sulphur and phosphorus.
Solution 33
The surface of some metals acquires a dull appearance when exposed to air for a long time because metals form a thin layer of oxides, carbonates or sulphide on their surface by the slow action of various gases present in air.
Solution 34
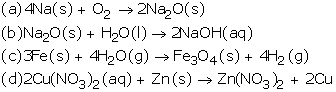
Solution 35
(a) Hydrogen.
(b) Magnesium oxide.
(c) Aluminium oxide.
(d) Sodium; Diamond.
(e) Less.
Solution 36
(a) Metals are malleable i.e. they can be beaten into thin sheets with a hammer.
Example: Aluminium.
Metals are ductile i.e. they can be drawn into thin wires.
Example: Copper.
(b) Aluminium and copper.
(c) Malleability.
(d) Ductility.
Solution 37
Sodium and potassium metals react violently with cold water.
Observations:
1. Metal starts moving over the surface of water making a hissing sound.
2. Metal starts reacting with water causing little explosions.
3. Soon the metal catches fire and starts burning.
If the gas evolved burns with a 'pop' sound, it indicates that it is hydrogen gas.
Solution 38
(a) Metal oxides are basic in nature and turn red litmus blue. For example: Magnesium oxide.
Non-metal oxides are acidic or neutral in nature. The acidic oxides turn blue litmus red. For example: Carbon dioxide.
(b) (i) Acidic oxide: S, C
(ii) Basic oxide: Na, K
(iii) Neutral oxide: H
Solution 39
(a) Those metal oxides which show basic as well as acidic behavior are known as amphoteric oxides.
Example: Aluminium oxide and zinc oxide.
(b) Acidic oxide - CO2, CO, SO2 and N2O; Basic oxide - Na2O, MgO; Neutral oxide - H2O.
(c) ZnO, Al2O3.
Solution 40
(a) SO2 is acidic in nature. When dissolved in water, it produces sulphurous acid.
(b) Na2O is basic in nature. When dissolved in water, it forms an alkali called sodium hydroxide.
Solution 41
(a) When non-metals react with oxygen, they form acidic oxides or neutral oxides.
Example: Carbon reacts with oxygen to form an acidic oxide called carbon dioxide. Hydrogen reacts with oxygen to form neutral oxide called water.
(b) When metals combine with oxygen, they form basic oxides.
Example: Sodium reacts with oxygen to form a basic oxide called sodium oxide.
Solution 42
(a) Metals do not liberate hydrogen gas with nitric acid because nitric acid is a strong oxidizing agent. So, as soon as hydrogen gas is formed in the reaction between a metal and dilute nitric acid, the nitric acid oxidises this hydrogen to water.
(b) Magnesium and manganese.
Solution 43
(a) Most of the metals do not react with hydrogen. Only a few reactive metals react with hydrogen to form metal hydrides. For example: When hydrogen gas is passed over heated sodium, then sodium hydride is formed.
(b) Non-metals react with hydrogen to form covalent hydrides. For example: Sulphur combines with hydrogen to form hydrogen sulphide.
Solution 44
(a) Calcium reacts vigorously with chlorine to form an ionic chloride called calcium chloride.
![]()
(b) Magnesium reacts with very dilute nitric acid to form magnesium nitrate and hydrogen gas.
![]()
Solution 45
(a) Sodium > Calcium > Magnesium > Zinc > Iron > Lead > Copper
(b) When a strip of zinc metal is put in copper sulphate solution, then the blue colour of copper sulphate solution fades gradually and red brown coating of copper is deposited on the zinc strip.
![]()
Metals and Non-metals Exercise 134
Solution 46
Silver gets deposited on the copper plate because copper is more reactive than silver and hence displaces silver from silver nitrate solution.
Solution 47
Uses of metals:
(i) Lead metal is used in making car batteries.
(ii) Zinc is used for galvanizing iron to protect it from rusting.
(iii) Iron, copper and aluminium are used to make utensils.
(iv) Copper and aluminium metals are used to make electrical wires.
(v) Aluminium is used to make aluminium foil for packaging materials.
Uses of non-metals:
(i) Hydrogen is used in the hydrogenation of vegetable oils.
(ii) Carbon is used to make electrodes of electrolytic cells and dry cells.
(iii) Nitrogen is used in the manufacture of ammonia, nitric acid and fertilizers.
(iv) Sulphur is used for producing sulphuric acid.
(v) Liquid hydrogen is used as rocket fuel.
Solution 48
(i) Copper - Copper is used to make wires to carry electric current.
(ii) Aluminium - Aluminium foils are used in packaging of food materials.
(iii) Iron - Iron is used to make utensils.
(iv) Silver - Silver is used to make jewellery.
(v) Gold - Gold is used to make jewellery.
(vi) Mercury - Mercury is used in thermometers.
Solution 49
(a) (i) Hydrogen - Hydrogen is used in the hydrogenation of vegetable oils.
(ii) Carbon is used to make electrodes of electrolytic cells and dry cells.
(iii) Nitrogen is used in the manufacture of ammonia, nitric acid and fertilizers.
(iv) Sulphur is used in making sulphuric acid.
(b) Mercury
Solution 50
(a) Aluminium metal does not react with water under ordinary conditions because of the presence of a thin layer of aluminium oxide on its surface.
(b) Sodium and magnesium.
(c) Copper and silver.
Solution 51
(a) Sodium is a very reactive metal so it reacts vigorously with the oxygen of air and catches fire. It is kept immersed in kerosene oil to protect it from the action of oxygen, moisture and carbon dioxide of air and to prevent accidental fires.
(b) White phosphorus is kept immersed in water because it reacts spontaneously with oxygen of air to form phosphorus pentoxide but does not react with water.
(c) No, because sodium reacts vigorously with water to form sodium hydroxide and hydrogen.
Solution 52
(a) Potassium reacts violently with cold water to form potassium hydroxide and hydrogen gas.
![]()
(b) ![]()
(c) Hydrogen.
Solution 53
(a) Magnesium reacts with very dilute nitric acid to form magnesium nitrate and hydrogen gas.![]()
(b) Magnesium and aluminium.
Solution 54
(a) Sodium reacts vigorously with cold water forming sodium hydroxide and hydrogen gas.
![]()
(b) Magnesium reacts with hot water to form magnesium hydroxide and hydrogen.
![]()
(c) Aluminium reacts with steam to form aluminium oxide and hydrogen gas.
![]()
Solution 55
(i) When sodium, magnesium and copper are left in air, sodium reacts vigorously with oxygen to form sodium oxide, magnesium reacts with oxygen to form magnesium oxide only on heating, whereas copper does not burn in air even on strong heating. It reacts only on prolonged heating. This shows that sodium is most reactive, then magnesium and copper is the least reactive among the three.
(ii) Sodium reacts vigorously with cold water to form sodium hydroxide and hydrogen, magnesium does not react with cold water but reacts with hot water to form magnesium hydroxide and hydrogen but copper does not react even with steam. This shows that sodium is highly reactive; magnesium is less reactive than sodium and copper is the least reactive among the three.
Solution 56
(a) ![]()
In this reaction, aluminium oxide behaves as a basic oxide because it reacts with an acid to form salt and water.
![]()
In this reaction, aluminium oxide behaves as an acidic oxide because it reacts with a base to form salt and water.
(b) Amphoteric oxides.
(c) Zinc oxide.
Solution 57
(a) Calcium reacts with cold water to form calcium hydroxide and hydrogen gas.
![]()
(b) When iron reacts with dilute sulphuric acid, it forms iron sulphate and hydrogen gas.
![]()
When hydrogen gas is ignited with a burning matchstick, it produces a 'pop' sound.
Solution 58
We would create an apparatus using dry cell, a torch bulb fitted in a holder and some connecting wires with crocodile clips and connect them to make an electric circuit. Then insert a piece of sulphur between the crocodile clips and the bulb does not light up at all. This means that sulphur does not allow the electric current to pass through it. Now insert a piece of copper between the crocodile clips and the bulb will light up. This observation shows that non metals (ex- sulphur) do not conduct electricity and metals (ex- copper) conduct electricity.
Solution 59
Properties of metals:
(i) Metals are malleable i.e. they can be beaten into thin sheets with a hammer.
(ii) Metals are ductile i.e. they can be drawn into thin wires.
(iii) Metals are good conductors of heat and electricity.
(iv) Metals are lustrous.
(v) Metals are generally hard.
Properties of non-metals:
(i) Non-metals are non-malleable i.e. they cannot be beaten into thin sheets with a hammer.
(ii) Non-metals are non-ductile i.e. they cannot be drawn into thin wires.
(iii) Non-metals are bad conductors of heat and electricity.
(iv) Non-metals are non-lustrous.
(v) Non-metals are generally soft.
Solution 60
(a) Sodium metal: Soft, low melting point
Carbon non-metal: graphite conducts electricity; diamond has a very high melting point.
(b) Gallium and cesium.
Solution 61
Metals lose their shine or brightness on keeping in air for a long time and acquire a dull appearence due to the formation of a thin layer of oxide, carbonate or sulphide on their surface by the slow action of various gases present in air.
Brightness of metals can be restored by rubbing the dull surface of the metal object with a sand paper, then the outer corroded layer is removed and the metal object becomes shiny and bright once again.
Solution 62
(a) Metals are the elements that conduct heat and electricity, and are malleable and ductile.
Example: Iron, aluminium, copper, gold and silver.
(b) Sodium
(c) Silver
(d) When a metal reacts with dilute hydrochloric acid, it forms metal chloride and hydrogen gas.
Example: Magnesium reacts rapidly with dilute hydrochloric acid to form magnesium chloride and hydrogen.
![]()
(e) (i) ![]()
The products formed are magnesium chloride and hydrogen.
(ii) ![]()
The products formed are aluminium chloride and hydrogen.
(iii) ![]()
The products formed are zinc chloride and hydrogen.
(iv) ![]()
The products formed are iron chloride and hydrogen.
Metals and Non-metals Exercise 135
Solution 63
(a) Non-metals are the elements that do not conduct heat and electricity and are neither malleable nor ductile.
Example: Carbon, sulphur, phosphorus, silicon and oxygen.
(b) Carbon.
(c) Iodine.
(d) Carbon (Diamond).
(e) Non-metals react with oxygen to form acidic oxides or neutral oxides. Carbon burns in air to form carbon dioxide.
![]()
The nature of the product formed is acidic. When carbon dioxide dissolves in water, it forms carbonic acid. It turns blue litmus to red which shows it is acidic in nature.
Solution 64
(a) The arrangement of metals in a vertical column in the order of decreasing reactivities is called reactivity series.
Increasing order of reactivity: Iron < zinc < magnesium < sodium
(b) Though hydrogen is not a metal but it has been placed in the reactivity series of metals due to the fact that like metals, hydrogen also loses electrons and forms positive ions.
(c) Lead is more reactive than hydrogen and copper is less reactive than hydrogen.
(d) Zinc displaces copper from copper sulphate solution and mercury does not displace copper from copper sulphate solution.
(e) Copper displaces silver from silver nitrate solution and gold does not.
Solution 65
(a) Difference between metals and non-metals:
|
Metals |
Non-metals |
|
(i) Metals are malleable i.e. they can be beaten into thin sheets with a hammer.
(ii) Metals are ductile i.e. they can be drawn into thin wires. (iii) Metals are good conductors of heat and electricity.
|
(i) Non-metals are non-malleable i.e. they cannot be beaten into thin sheets with a hammer. (ii) Non-metals are non-ductile i.e. they cannot be drawn into thin wires. (iii) Non-metals are bad conductors of heat and electricity.
|
(b) Difference between metals and non-metals:
|
Metals |
Non-metals |
|
(i) Metals form basic oxides. (ii) Metals displace hydrogen from water (iii) Metals displace hydrogen from dilute acids. |
(i) Non-metals form acidic or neutral oxides. (ii) Non-metals do not react with water. (iii) Non-metals do not react with dilute acids. |
(c) Sodium is a solid, it conducts electricity and forms basic oxides.
(d) Sulphur is a non-metal as it is brittle, non-ductile, non-conductor of electricity and forms acidic oxides.
(e) Hydrogen.
Metals and Non-metals Exercise 136
Solution 89
(a) Basic oxide.
(b) Metal.
(c) Sodium, Na.
Solution 90
(a) Amphoteric oxide.
(b) Amphoteric oxide.
(c) Aluminium, Al.
(d) Zinc, Zn.
Solution 91
(a) Neutral oxide.
(b) Acidic oxide.
(c) X is non-metal because non-metals form acidic and basic oxide.
(d) Carbon, C.
Solution 92
(a) No displacement reaction will take place because gold is less reactive than copper.
(b) No reaction will take place between copper and copper sulphate solution; there is no reaction possible.
(c) Zinc displaces copper from copper sulphate solution to form zinc sulphate solution and copper metal because zinc is more reactive than copper.
(d) No displacement reaction will take place because mercury is less reactive than copper.
Solution 93
(a) Metal chloride: Sodium chloride, NaCl
Non-metal chloride: Carbon tetrachloride, CCl4
(b) Sodium chloride solution conducts electricity whereas carbon tetrachloride does not conduct electricity.
(c) Sodium chloride is an ionic compound whereas carbon tetrachloride is a covalent compound.
Metals and Non-metals Exercise 137
Solution 94
M is more reactive than lead since it is able to displace lead from lead acetate solution.
Solution 95
Zinc is most reactive and copper is least reactive out of the three since iron displaced copper from its solution and zinc displaced iron from its solution.
Solution 96
Reaction (a) will not occur because Cu is less reactive than Mg
Reaction (c) will also not occur because Fe is less reactive than Mg.
Solution 97
Metal B will be nearer to the top of the activity series since it is highly reactive and is hence found in the form of its compounds and not in free state.
Solution 98
K being the lowest in the reactivity series is least reactive and is most likely to occur in a free state in nature.
Solution 99
(a) (i) Iron (ii) Gold
(b) More heat is evolved during the reaction of sodium metal with water due to which the hydrogen gas formed catches fire. On the other hand, less heat is evolved during the reaction of calcium metal with water which cannot make the hydrogen gas burn.
Solution 100
Zinc metal is more reactive than copper. Some of the zinc metal of zinc plate dissolves and displaces copper from copper sulphate solution. This dissolving of zinc metal forms tiny holes in zinc plate. Blue colour of copper sulphate solution gets lighter and lighter due to the formation of colourless zinc sulphate solution.
Metals and Non-metals Exercise 167
Solution 1
(a) Covalent bond.
(b) Ionic bond.
Solution 2
Carbon dioxide, CO2
Solution 3
![]()
Solution 4
(a) Ionic bond.
(b) Covalent bond.
Solution 5
(a) Ions
(b) Anions
(c) Cations
Solution 6
(a) No. of electrons in Na+= 10
(b) No. of electrons in Cl- = 18
Solution 7
X will form X2-
Y will form Y2+
Solution 8
(a) (i) E.C of Mg = 2, 8, 2 (ii) E.C of Mg2+ = 2, 8
(b) (i) E.C of S = 2, 8, 6 (ii) E.C of S2- = 2, 8, 8
Solution 9
Ionic bonds.
Solution 10
False (It should be 'ions' in place of 'electrons').
Solution 11
Covalent bond
Solution 12
(i)
![]()
(ii)
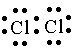
Solution 13
Covalent bonds are present in a water molecule.
Electron dot structure of water:
Solution 14
Methane: Covalent bonds
Sodium chloride: Ionic bonds
Solution 15
Ionic compounds conduct electricity when dissolved in water or melted whereas covalent compounds do not conduct electricity.
Ionic compound - NaCl
Covalent compound - CO2
Solution 16
Covalent bonds are present in the given molecules.
(i) H2
![]()
(ii) CH4
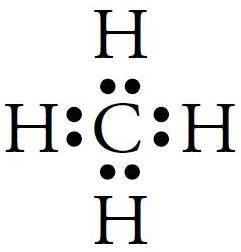
(iii) Cl2
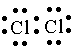
(iv) O2
![]()
Solution 17
Argon; E.C = 2,8,8
Solution 18
Ionic compounds: Sodium chloride, Ammonium chloride, Magnesium chloride.
Covalent compounds: Urea, Cane sugar, Hydrogen chloride, Carbon tetrachloride, Ammonia, Alcohol.
Solution 19
(i) Hydrogen
(ii) Oxygen
(iii) Nitrogen
(iv) Sodium chloride
Metals and Non-metals Exercise 168
Solution 20
(i) Covalent
(ii) Covalent
(iii) Two
(iv) Three
(v) Two
(vi) Higher
Solution 21
(a) The chemical bond formed by the sharing of electrons between two atoms is known as a covalent bond.
(i) Covalent bonds (ii) Ionic bonds
(b) The chemical bond formed by the transfer of electrons from one atom to another is known as an ionic bond.
Covalent bond is present in an oxygen molecule.
Solution 22
(a) An ion is an electrically charged atom (or group of atoms). Example: Sodium ion, Na+, magnesium ion, Mg2+.
(b) (i) Positive charge (ii) Negative charge
(c) Cation: Magnesium ion, Mg2+
Anion: Chloride ions, 2Cl-
Solution 23
(a) Covalent bond; Because whenever two atoms of the same element combine to form a molecule, a covalent bond is formed.
(b) The atomic number of chlorine is 17, so its electronic configuration is 2,8,7. Chlorine atom has 7 electrons in its outermost shell and needs 1 more electron to complete its octet and become stable. It gets this electron by sharing with another chlorine atom. So, two chlorine atoms share one electron each to form a chlorine molecule. Because the two chlorine atoms share electrons, there is a strong force of attraction between them which holds them together. This force is called covalent bond. Now, each chlorine atom in the chlorine molecule has the electronic configuration 2,8,8 resembling its nearest inert gas argon. Since the chlorine atoms in a chlorine molecule have inert gas electron arrangements, therefore, a chlorine molecule is more stable than two separate chlorine atoms.
Solution 24
(a) (i) The compounds containing ionic bonds are known as ionic compounds. They are formed by the transfer of electrons from one atom to another.
Example: Sodium chloride, NaCl.
(ii) The compounds containing covalent bonds are known as covalent compounds. They are formed by the sharing of electrons between atoms. Example: Methane, CH4.
(b)
|
Ionic compounds |
Covalent compounds |
|
(i) Ionic compounds are usually crystalline solids. (ii) Ionic compounds have high melting and boiling points. (iii) Ionic compounds are usually soluble in water. |
(i) Covalent compounds are usually liquids or gases. (ii) Covalent compounds have low melting and boiling points. (iii) Covalent compounds are usually insoluble in water. |
Solution 25
(a) Covalent compounds have generally low melting points because they are made up of electrically neutral molecules. So, the force of attraction between the molecules of a covalent compound is very weak. Hence, only a small amount of heat energy is required to break these weak molecular forces.
(b) Ionic compounds are made of up of positive and negative ions. There is a strong force of attraction between the oppositely charged ions, so a lot of heat energy is required to break this force of attraction and melt or boil the ionic compound. Due to this, ionic compounds have high melting points.
Solution 26
(a) Ionic compounds:
(i) They have high melting and boiling points.
(ii) They are usually soluble in water.
Covalent compounds:
(i) They have low melting and boiling points.
(ii) They are usually insoluble in water.
(b) An aqueous solution of sodium chloride conducts electricity but a sugar solution does not conduct electricity.
Solution 27
(a) Ionic compounds conduct electricity in solution because they are made up of electrically charged ions but covalent compounds are made up of electrically neutral molecules so they do not conduct electricity.
(b) Conduct electricity: MgCl2, NaCl, Na2S (Ionic compounds)
Do not conduct electricity: CCl4, CS2 (Covalent compounds).
Solution 28
(a) Ionic compound: Sodium chloride, NaCl.
Covalent compound: Carbon tetrachloride, CCl4
(b) Out of A and B, the compound whose aqueous solution conducts electricity will be an ionic compound.
Solution 29
Common salt is an ionic compound containing ionic bonds whereas cane sugar is a covalent compound containing covalent bonds. Since, ionic compounds conduct electricity and covalent compounds do not, hence common salt is a good conductor of electricity and cane sugar is a non-conductor of electricity.
Solution 30
(a) Mg3N2
(b) Li2O
(c) AlCl3
(d) KH
Solution 31
(a) There are some elements in group 18 of the periodic table which do not combine with other elements. These elements are helium, neon, argon, krypton, xenon and radon. They are known as noble gases or inert gases because they are unreactive.
If we look at the electronic configuration of noble gases, we would notice that except helium, all other inert gases have 8 electrons (helium has 2) in their outermost shells. This is considered to be the most stable arrangement of electrons.
(b) Atoms form chemical bonds to achieve stability by acquiring the inert gas electron configuration.
Solution 32
(i) Magnesium
![]()
Oxygen

(ii) Formation of MgO
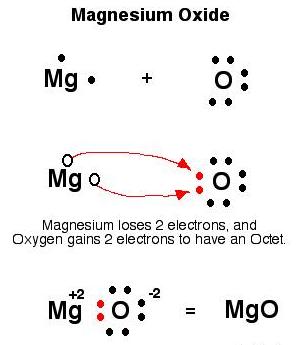
(iii) Both positive and negative ions i.e. Mg2+ and O2- respectively.
Solution 33
![]()
(i) Helium
(ii) Argon
Solution 34
(i) Ionic bonding.
(ii) Covalent bonding.
(iii) Covalent bonding.
Metals and Non-metals Exercise 169
Solution 35
The atomic number of sodium is 11, so its electronic configuration is 2,8,1. Sodium atom has only 1 electron in its outermost shell. So, the sodium atom donates one electron (to a chlorine atom) and forms a sodium ion, Na+. The atomic number of chlorine is 17, so its electronic configuration is 2,8,7. Chlorine atom has 7 electrons in its outermost shell and needs 1 more electron to achieve the stable 8-electron inert gas configuration. So, a chlorine atom takes one electron (from the sodium atom) and forms a negatively charged chloride ion, Cl- . This type of bonding is called ionic bonding.
Solution 36
A positively charged ion is known as cation. A cation is formed by the loss of one or more electrons by an atom. For example: sodium loses 1 electron to form a sodium ion, Na+, which is a cation.
A negatively charged ion is known as anion. An anion is formed by the gain of one or more electrons by an atom. For example: A chlorine atom gains (accepts) 1 electron to form a chloride ion, Cl-, which is an anion.
Solution 37
Since nitrogen has 5 electrons in its outermost shell so, to achieve the 8-electron structure of an inert gas, it needs 3 more electrons and hence combines with another nitrogen atom to form a molecule of nitrogen gas.
This type of bonding is called covalent bonding.
Solution 38
(i) CO2 - Covalent bond
![]()
(ii) MgO - Ionic bond
![]()
(iii) H2O - Covalent bond
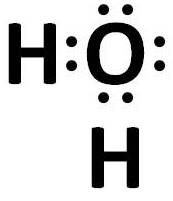
(iv) HCl - Covalent bond
![]()
(v) MgCl2 - Ionic bond
![]()
Solution 39
Since an oxygen atom has 6 electrons in its outermost shell so, it needs 2 more electrons to achieve the stable 8-electron inert gas configuration. Hence, it combines with another oxygen atom and forms a molecule of oxygen.
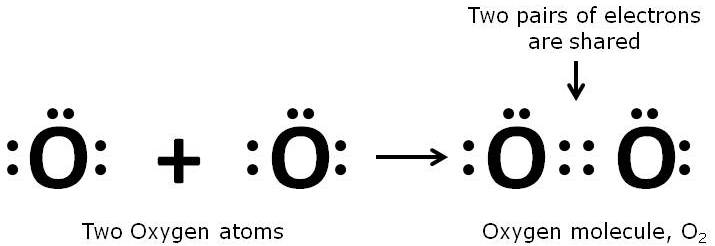
This type of bonding is called a double covalent bond.
Solution 40
(i) KCl - Ionic bond
![]()
(ii) NH3 - Covalent bond
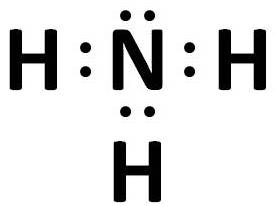
(iii) CaO - Ionic bond
![]()
(iv) N2 - Covalent bond
![]()
(v) CaCl2 - Ionic bond
![]()
Solution 41
Although solid ionic compounds are made up of ions but they do not conduct electricity in solid state. This is because in the solid ionic compound, the ions are held together in fixed positions by strong electrostatic forces and cannot move freely. However, when we dissolve the ionic solid in water or melt it, the crystal structure is broken down and ions become free to move and conduct electricity. Thus, an aqueous solution of an ionic compound conducts electricity because there are plenty of free ions in the solution which are able to conduct electric current.
Solution 42
(a) (i) Sodium - 2, 8, 1 (ii) Chlorine - 2, 8, 7
(b) (i) Sodium = 1 (ii) Chlorine = 7
(c)
(d) Sodium chloride has a high melting point because it is an ionic compound and these compounds are made of up of positive and negative ions. There is a strong force of attraction between the oppositely charged ions, so, a lot of heat energy is required to break this force of attraction and melt or boil the ionic compound.
(e) Anode: Thick block of impure copper metal; Cathode: Thin strip of pure copper metal.
Solution 43
(a) (i) Magnesium - 2, 8, 2 (ii) Oxygen - 2, 6
(b) (i) Magnesium = 2 (ii) Oxygen = 6
(c)
(d) Water.
(e) An aqueous solution of an ionic compound conducts electricity because there are plenty of free ions in the solution which are able to conduct electric current.
Solution 44
(a) (i) 2,8,1 (ii) 2,6
(b) (i) 1 (ii) 6
(c)
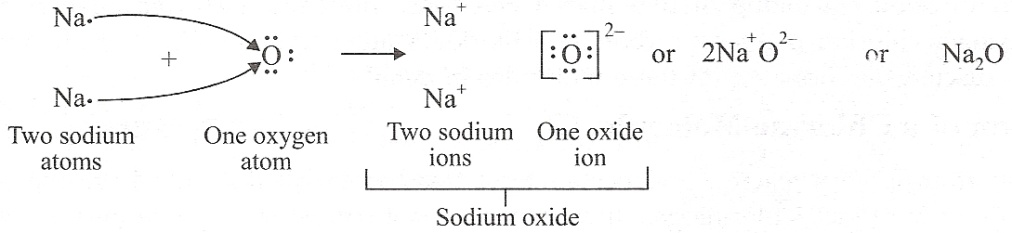
(d) Ionic compounds are usually hard because their oppositely charged ions attract one another strongly and form a regular crystal structure.
(e) Although solid ionic compounds are made up of ions but they do not conduct electricity in solid state. This is because in the solid ionic compound the ions are held together in fixed positions by strong electrostatic forces and cannot move freely. However, when we dissolve the ionic solid in water or melt it, the crystal structure is broken down and ions become free to move and conduct electricity. Thus, an aqueous solution of an ionic compound conducts electricity because there are plenty of free ions in the solution which are able to conduct electric current.
Solution 45
(a) (i) Magnesium: 2, 8, 2 (ii) Chlorine: 2, 8, 7
(b) (i) 2 (ii) 7
(c)
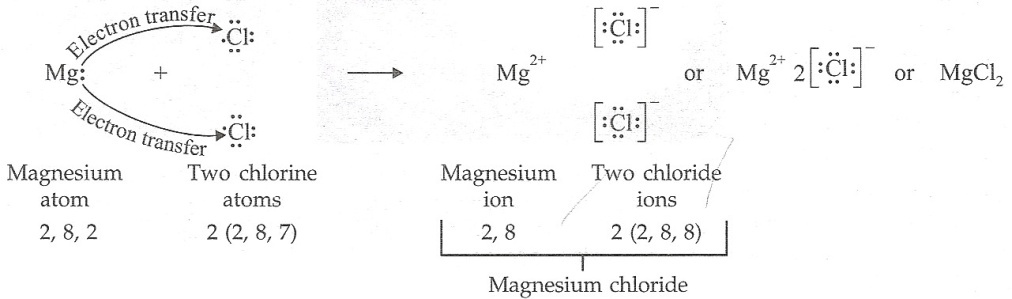
(d) Magnesium chloride will conduct electricity because it is an ionic compound.
(e) Covalent compounds are generally poor conductors of electricity because they do not contain ions.
Metals and Non-metals Exercise 171
Solution 67
(a) Covalent bond
(b) Low melting point
(c) No
(d) Yes
Solution 68
(i) Ionic bond
(ii) High melting point and boiling point
(iii) Yes
(iv) No
Solution 69
(a) Y and Z will form an ionic compound because an ionic bond is formed when one of the atoms can donate electrons to achieve the inert gas configuration, and the other atom needs electrons to achieve the inert gas configuration. Since Y has 7 and Z has 1 electron in their outermost shell, they would form an ionic bond.
(b) X and Y will react to form a covalent compound because a covalent bond is formed when both the reacting atoms need electrons to achieve the inert gas electron arrangement.
Solution 70
Covalent bond will be formed since covalent compounds are non conductors of electricity.
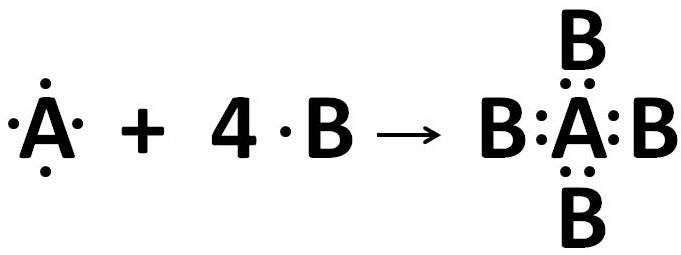
Solution 71
XY2 has ionic bonds.
(i) XY2 would conduct electricity when dissolved in water.
(ii) XY2 would have high melting and boiling point.
Solution 72
AB2 forms ionic bonds because an aqueous solution of an ionic compound conducts electricity because there are plenty of free ions in the solution which are able to conduct electric current.
Solution 73
(a) Covalent bond.
(b) Ionic bond.
Solution 74
(a) Ionic bond will be formed; element C donates its electron to element D.
(b) B is an inert gas; has complete octet configuration.
(c) C2A; element A needs two electrons to complete its octet.
Solution 75
XY2 forms ionic bond. The electronic configuration of X changes from 2, 8, 2 to 2, 8, it donates its 2 electrons to two Y atoms. Hence, the electronic configuration of Y changes from 2, 8, 7 to 2, 8, 8 giving the compound XY2.
Solution 76
(a) A is a metal
(b) B and C are non-metals
(c) A and B combines to form an ionic bond.
(d) B and C combines to form a covalent bond.
(e) B will form an anion with valency 1 since it needs only 1 electron to complete its octet.
Metals and Non-metals Exercise 172
Solution 77
(i) B: Magnesium atom (At. no. = 12)
(ii) C: Oxygen atom (At. no. = 8)
(iii) D: Sodium ion (E.C = 2, 8)
(iv) A: Chloride ion (E.C = 2, 8, 8)
Solution 78
(a) An atom of X loses 2 electrons to attain the nearest gas electron configuration (2,8).
(b) Neon
Solution 79
(a) An atom of Y accepts 2 electrons to achieve the nearest inert gas electron arrangement (2, 8, 8).
(b) Argon
Solution 80
Solid air-fresheners are covalent compounds because they are volatile compounds with low melting and boiling points.
Solution 81
Formula of chloride of element X is XCl
Formula of chloride of element Y is YCl4
The properties of two chlorides will be different because XCl is an ionic chloride whereas YCl4 is a covalent chloride.
Metals and Non-metals Exercise 191
Solution 1
Calamine, ZnCO3
Solution 2
Reduction.
Solution 3
Rusting of iron can be prevented:
(i) By painting.
(ii) By applying grease or oil.
Solution 4
The process of depositing a thin layer of zinc metal on iron objects is called galvanisation; It prevents iron from rusting.
Solution 5
Zinc is used for galvanising iron.
Solution 6
Zinc is a quite reactive metal. The action of air on zinc metal forms a very thin coating of zinc oxide all over it, which is hard and impervious to air and hence prevents the further corrosion of zinc metal as well as the iron below it.
Solution 7
Iron objects are painted so that air and moisture can not come in contact with the iron objects and hence no rusting takes place.
Solution 8
Carbonate and sulphide ores are usually converted into oxides because it is easier to obtain metals from their oxides (by reduction) than from carbonates or sulphides.
Solution 9
Aluminium powder is used as the reducing agent for the extraction of manganese from its oxide.
Solution 10
Solder is an alloy of lead and tin.
Solution 11
Solder is an alloy of lead (Pb) and tin (Sn). It contains both the elements in 50-50 ratio. It has a low melting point and is used for soldering electrical wires together.
Solution 12
An alloy of mercury metal with one or more other metals is known as an amalgam.
Solution 13
Pure gold is said to be of 24 carats. It is not suitable for making ornaments because it is very soft.
Solution 14
Electrolytic refining.
Solution 15
(i) Presence of air (oxygen).
(ii) Presence of water (or moisture).
Solution 16
Rusting of iron can be prevented by alloying iron with chromium and nickel to make stainless steel.
Solution 17
Steel - Iron and carbon.
Stainless steel - Iron, chromium and nickel.
Solution 18
Silver, gold and platinum are used to make jewellery because all of these metals have a bright shiny surface and are resistant to corrosion.
Solution 19
Silver metal becomes black in the presence of hydrogen sulphide gas in air.
Solution 20
Hydrogen sulphide gas tarnishes silver articles.
Solution 21
The silver articles combine slowly with the hydrogen sulphide gas present in air to form a black coating of silver sulphide. The tarnishing of the silver objects is due to this silver sulphide coating on the object's surface.
Solution 22
Bronze is an alloy of copper and tin; 90% copper and 10% tin. It is used for making statues and coins.
Solution 23
A new aluminium vessel lose shine so soon after use due to the corrosion of aluminium metal when exposed to moist air. This happens because the oxygen of air reacts with aluminium to form a thin, dull layer of aluminium oxide all over the vessel.
Solution 25
Gold and platinum are highly resistant to corrosion.
Solution 26
Low melting point of solder makes it sutaible for welding electrical wires.
Solution 27
Carbon cannot reduce oxides of sodium or magnesium because carbon is less reactive than magnesium or sodium. Carbon, which is a non-metal, is more reactive than zinc and can be placed just above Zn in the reactivity series. Hence, carbon can reduce the oxides of zinc and all other metals below zinc to form metals.
Solution 28
The metals like Na, K, Ca and Mg never found in their free state in nature because of the reason that all of these metals are high-up in the reactivity series. And just because they are so reactive, they are never found in nature as free elements.
Solution 29
(a) Zinc
(b) Sodium
(c) Manganese
(d) Mercury
Solution 30
(a) rusting
(b) air; water
(c) galvanisation
(d) tin; chromium
(e) green
(j) zinc
(g) tin
(h) carbon
(i) amalgam
(j) less
(k) gangue
Solution 24
Metals and Non-metals Exercise 192
Solution 31
Manganese metal is extracted by the reduction of its oxide with aluminium powder as the reducing agent. Thus, when manganese dioxide is heated with aluminium powder, then manganese metal is formed.
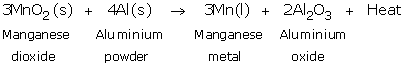
Solution 32
The reduction of a metal oxide to form metal by using aluminium powder as a reducing agent is called a thermite reaction.
This property of reduction by aluminium is made use of in thermite welding for joining the broken pieces of heavy iron objects like girders etc.
A mixture of Iron (III) oxide and aluminium powder is ignited with a burning magnesium ribbon. Aluminium reduces iron oxide to produce iron metal with the evolution of a lot of heat. Due to this heat, iron metal is produced in the molten state. This molten iron is then poured between the broken iron pieces to weld them (to join them).
![]()
Solution 33
Electrolytic reduction: Aluminium and Sodium;
Reduction with carbon : Zinc, Iron and Tin;
Reduction with aluminium: Manganese
Solution 34
(a) Copper does not corrode easily in the presence of water but steel rusts in the presence of water.
(b) The surface of some metals acquires a dull appearance when exposed to air because of the formation of an oxide layer on the surface of the metal.
Solution 35
(a) Aluminium does not corrode right through because aluminium is more reactive than iron and it forms a layer of aluminium oxide as soon as it comes in contact with moist air. This aluminium oxide layer is very tough and prevents the aluminium underneath from corroding.
(b) The process of thickening of aluminium oxide layer on the surface of aluminium objects by electrolysis is called anodizing. It is done to provide more protection to the aluminium object from corrosion.
Solution 36
(a) An iron grill is painted frequently to prevent its rusting.
(b) There is less corrosion of aluminium than iron when both are exposed to air because aluminium forms a layer of aluminium oxide on its surface as soon as it comes in contact with moist air. This aluminium oxide is very tough and prevents it from corroding right through.
Solution 37
(a) Electrolytic reduction.
(b) Copper glance (Cu2S)
(c) When calamine ore is heated strongly in the absence of air i.e. calcined, it decomposes to form zinc oxide and carbon dioxide.
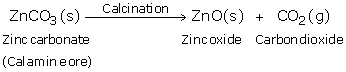
Then, zinc oxide is heated with carbon and zinc metal is produced.
![]()
Solution 38
(a) Copper and Silver occur in nature in free state as well as in combined state.
(b) Pyrolusite; Manganese dioxide; MnO2
(c)
(i) Roasting: When zinc sulphide (zinc blende ore) is strongly heated in air (roasted), it forms zinc oxide and sulphur dioxide.
(ii) Reduction: Zinc oxide obtained is heated with carbon to form zinc metal.
Solution 39
Different methods are used for extracting metals belonging to category of highly reactive metals, moderately reactive metals and less reactive metals. This is because the extraction of a metal from its concentrated ore is essentially a process of reduction of the metal compound present in the ore. For example: Manganese metal is obtained by the reduction of its oxide with aluminium powder and not carbon. This is because carbon is less reactive than manganese. Carbon, which is a non-metal, is more reactive than zinc and it can be placed just above Zn in the reactivity series. Hence, carbon can reduce the oxides of zinc and all other metals below zinc to form metals
Solution 40
The highly reactive metals are extracted by the electrolytic reduction of their molten chlorides or oxides.
Example: Sodium metal is extracted by the electrolytic reduction of molten sodium chloride. When electric current is passed through molten sodium chloride, it decomposes to form sodium metal and chlorine gas.
![]()
Solution 41
The moderately reactive metals are extracted by the reduction of their oxides with carbon, aluminium, sodium or calcium.
Example: When zinc sulphide (zinc blende ore) is strongly heated in air (roasted), it forms zinc oxide and sulphur dioxide. This process is called roasting. Then, zinc oxide is heated with carbon to form zinc metal. This process is termed as reduction.
Solution 42
The less reactive metals are extracted by the reduction of their oxides by heat alone.
Example: Mercury (II) sulphide ore is roasted in air when mercury (II) oxide is formed. When this mercury (II) oxide is heated to about 300oC, it decomposes to form mercury metal.
Solution 43
The process of purifying impure metals is called refining of metals.
Electrolytic refining is the most widely used method for the refining of impure metals obtained by various reduction processes.
In an electrolytic tank, acidified copper sulphate (CuSO4 + dilute H2O4) solution forms the electrolyte. A block of impure copper is made into an anode by connecting the positive terminal of a power supply (battery). A thin strip of highly pure copper metal is the cathode of the cell. The negative terminal of the power supply is connected to it.
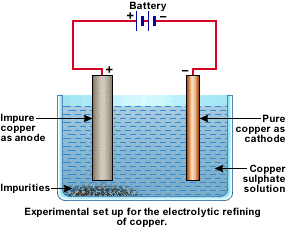
A small electric current is passed through the cell. Atoms from the anode enter the electrolyte. The copper from the anode gets converted into copper sulphide. An equal number of copper atoms from the solution get deposited on the cathode. This is to keep the concentration of the solution constant. Impurities from the anode block either remain in solution or collect below the anode, as they are unable to displace copper from the sulphate solution. The insoluble impurities remain in the electrolyte and are called anode mud.
Copper sulphate solution contains ions of Cu++ and SO4--. The following reactions take place at the anode and cathode when an electric current is passed.
![]()
![]()
Pure copper is scraped or removed from the cathode. Anode becomes thinner as the electrolysis process proceeds. Some important metals like gold and silver are present in the anode mud. These can be recovered separately.
Solution 44
(a) (i) Minerals - The natural materials in which the metals or their compounds are found in earth are called minerals.
(ii) Ores - Those minerals from which the metals can be extracted conveniently and profitably are called ores.
(iii) Gangue - The unwanted impurities like sand, rocky material, earthy particles etc., present in an ore are called gangue.
(b) Before extracting metal from an ore, it is necessary to remove these impurities (gangue) from it. By removing the gangue, we get a concentrated ore containing a much higher percentage of metal. This is called concentration of ore; also known as enrichment of ore.
(c) Ore: Copper glance; Copper (I) sulphide, Cu2S.
Metals and Non-metals Exercise 193
Solution 45
A mixture of Iron (III) oxide and aluminium powder is ignited with a burning magnesium ribbon. Aluminium reduces iron oxide to produce iron metal with the evolution of lot of heat. Due to this heat, iron metal is produced in the molten state. This molten iron is poured between broken iron parts of the machine to weld them (to join them).
![]()
Solution 46
(a) The eating up of metals by the action of air, moisture or a chemical (such as an acid) on their surface is called corrosion.
(b) Gold and Platinum
(c) Rusting
(d) Aluminium begins to corrode quickly when it comes in contact with moist air. The action of moist air on aluminium metal forms a thin layer of aluminium oxide all over the metal. This aluminium oxide is very tough and prevents the metal underneath from further corrosion. Therefore, aluminium is used for making utensils irrespective of its highly reactive property as its corrosion leads to the non-corrosion of the metal in the longer run.
Solution 47
When an iron object is left in damp air (or water) for a considerable time, it gets covered with a red-brown flaky substance called rust. This is called rusting of iron.
Experiment to show that rusting of Iron requires both, air and water:
We take three test-tubes and put one clean iron nail in each of the three test-tubes:
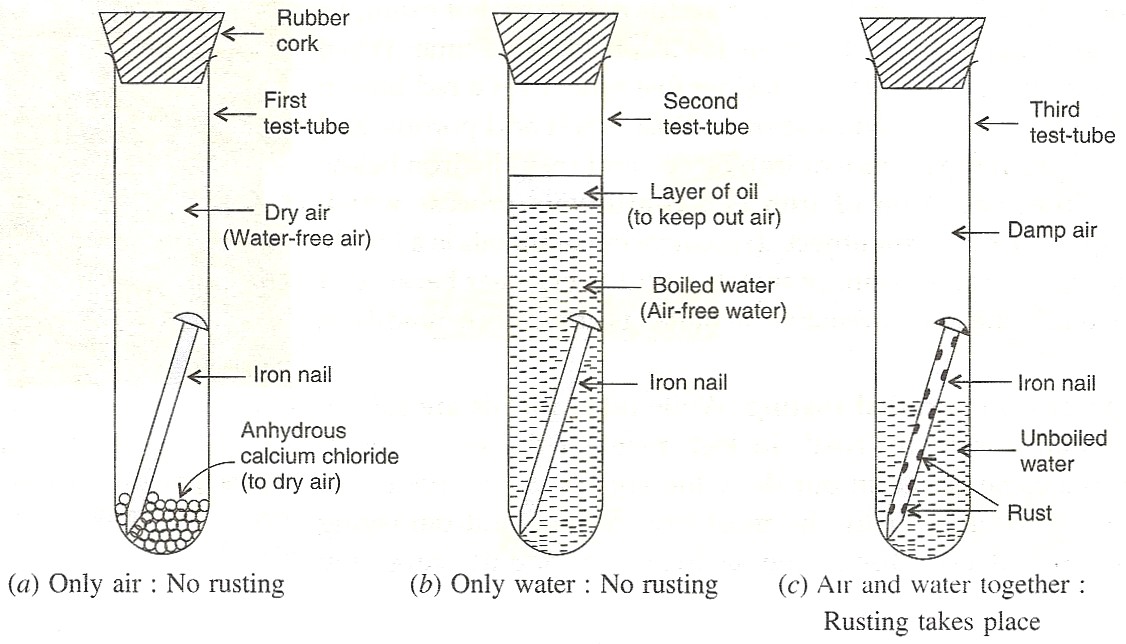
1. In the first test-tube containing iron nail, we put some anhydrous calcium chloride and close its mouth with a tight cork. Anhydrous calcium chloride absorbs water moisture from the damp air present in the test-tube and make it dry. In this way, the iron nail in the first test-tube is kept in dry air (having no water vapour in it).
2. In the second test-tube containing iron nail, we put boiled distilled water. Boiled water does not contain any dissolved air (or oxygen) in it (this is because the process of boiling removes all the dissolved air from it). A layer of oil is put over boiled water in the test-tube to prevent the outside air from mixing with boiled water. In this way, the iron nail in the second test-tube is kept in air free boiled water.
3. In the third test-tube containing an iron nail, we put unboiled water so that about two-thirds of nail is immersed in water and the rest is above the water, exposed to damp air. In this way, the iron nail in the third test-tube has been placed in air and water together.
The mouth of all three test tubes is closed with a cork and it is kept aside for about one week.
After one week, we observe the iron nails kept in all the three test-tubes, one by one. We find that (i) No rust is seen on the surface of iron nail kept in dry air (water-free air) in the first test-tube. This tells us that rusting of iron does not take place in air alone.
(ii) No rust is seen on the surface of iron nail kept in air-free, boiled water in the second test-tube. This tells us that rusting of iron does not take place in water alone.
(iii) Red-brown rust is seen on the surface of iron nail kept in the presence of both air and water together the third test-tube. This tells us that rusting of iron takes place in the presence of both air and water together.
Solution 48
(a) An alloy is a homogeneous mixture of two or more metals (or a metal and small amount of non-metals). An alloy is prepared by mixing the various metals in molten state in required proportions, and then cooling their mixture to the room temperature.
(b) Steel contains iron and carbon.
This alloy of iron (steel) is hard and strong. It also rusts less readily than pure iron.
(c) Brass contains copper and zinc.
Brass is used for making cooking utensils.
Solution 49
(a) Aluminium and Zinc resist corrosion due to the formation of a thin, hard and impervious layer of oxide on their surface.
(b) (i) Painting (ii) Applying grease or oil (iii) Galvanisation (iv) Tin and chromium plating (v) Alloying to form stainless steel.
(c) Stainless steel contains iron, chromium and nickel.
Stainless steel does not rust at all and is strong and tough.
Solution 50
(a) Brass: It contains Copper (Cu) - 80% and Zinc (Zn) - 20%. It is used for making cooking utensils.
(b) When a copper object remains in damp air for a considerable time, then copper reacts slowly with the carbon dioxide and water of air to form a green coating of basic copper carbonate on the surface of the object. The formation of this green coating of basic copper carbonate corrodes it. This process is known as corrosion of copper.
Solution 51
(a) When a coat of paint is applied to the surface of an iron object, it prevents air and moisture to come in contact with the object; hence no rusting takes place.
(b) The electrical conductivity of copper alloys like brass and bronze is less than that of pure copper.
(c) It means that 22 parts pure gold is alloyed with 2 parts of either silver or copper for making ornaments; Silver and copper are usually alloyed with gold to make it harder.
Solution 52
(a) When zinc carbonate is heated strongly in the absence of air, it decomposes to form zinc oxide and carbon dioxide.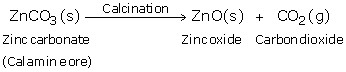
(b) When copper (I) oxide reacts with copper (I) sulphide, it forms copper metal and sulphur dioxide.
Solution 53
(a) Aluminium can be used a reducing agent other than carbon.
(b) We cannot use an aqueous solution of sodium chloride to obtain sodium metal because if we electrolyse an aqueous solution of sodium chloride, then as soon as sodium metal is produced at cathode, it will react with water present in the aqueous solution to form sodium hydroxide. Hence, electrolysis of an aqueous solution of sodium chloride will produce sodium hydroxide and not sodium metal.
Solution 54
For the refining of an impure metal by the process of electrolysis, a thick block of impure metal is made anode (connected to +ve terminal of the battery) and a thin strip of the pure metal is made cathode (connected to -ve terminal of battery). A water soluble salt (of the metal to be refined) is taken as electrolyte. On passing current, impure metal dissolves from the anode and goes into the electrolyte solution. And pure metal from the electrolyte deposits on the cathode.
Electrolytic refining of copper: In an electrolytic tank, acidified copper sulphate (CuSO4 + dilute H2O4) solution forms the electrolyte. A block of impure copper is made into an anode by connecting the positive terminal of a power supply (battery). A thin strip of highly pure copper metal is the cathode of the cell. The negative terminal of the power supply is connected to it.
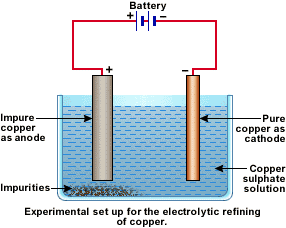
A small electric current is passed through the cell. Atoms from the anode enter the electrolyte. The copper from the anode gets converted into copper sulphide. An equal number of copper atoms from the solution get deposited on the cathode. This is to keep the concentration of the solution constant. Impurities from the anode block either remain in solution or collect below the anode, as they are unable to displace copper form the sulphate solution. The insoluble impurities remain in the electrolyte and are called anode mud.
Copper sulphate solution contains ions of Cu++ and SO4--. The following reactions take place at the anode and cathode when an electric current is passed.
![]()
![]()
Pure copper is scraped or removed from the cathode. Anode becomes thinner as the electrolysis process proceeds. Some important metals like gold and silver are present in the anode mud. These can be recovered separately.
Solution 55
(a) Aluminium oxide is electrolysed in molten state to obtain aluminium metal. Oxygen gas is evolved during the process.
(b) Sodium chloride is electrolysed in molten state to obtain sodium metal. Chlorine gas is evolved during this process.
(c) Carbon dioxide is produced when calamine ore is calcined.
(d) Sulphur dioxide gas is evolved when cinnabar ore is roasted.
Solution 56
(a) Gold and Platinum are found in nature mainly in the free state.
(b) Sodium and Magnesium are always found in combined state.
(c) Iron (III) oxide; Fe2O3 is present in haematite ore.
Solution 57
(a) The natural materials in which the metals or their compounds are found in earth are called minerals. Those minerals from which the metals can be extracted conveniently and profitably are called ores.
(b) Mercury.
(c) Rock salt - Sodium chloride, NaCl.
(d) Sodium metal is extracted by the electrolytic reduction of molten sodium chloride. When electric current is passed through molten sodium chloride, it decomposes to form sodium metal and chlorine gas.
![]()
(e) Potassium, Calcium and Magnesium.
Solution 58
(a) Iron is extracted from haematite ore.
(b) Bauxite; Aluminium oxide, Al2O3.2H2O
(c) Aluminium metal is extracted by the electrolytic reduction (electrolysis) of molten aluminium oxide. When electric current is passed through molten aluminium oxide, it decomposes to form aluminium metal and oxygen gas.
![]()
(d) Aluminium metal is produced at Cathode (Negative electrode).
(e) Oxygen gas is produced; at anode (Positive electrode).
Metals and Non-metals Exercise 194
Solution 59
(a) Aluminium.
(b) Haematite; Iron (III) oxide, Fe2O3
(c) Zinc sulphide (zinc blende ore) is strongly heated in air (roasted), it forms zinc oxide and sulphur dioxide. This process is called roasting.
Then, zinc oxide is heated with carbon to form zinc metal. This process is termed as reduction.
![]()
(d) The galvanized iron object remains protected against rusting even if a break occurs in the zinc layer because zinc is more easily oxidised than iron. Hence, the zinc continues to corrode but iron object does not corrode or rust.
(e) Aluminium.
Solution 60
(a) Sodium.
(b) (i) Calamine; Zinc carbonate, ZnCO3
(ii) Zinc blende; Zinc sulphide, ZnS
(c) Mercury (II) sulphide ore is roasted in air when mercury (II) oxide is formed.
When this mercury (II) oxide is heated to about 300oC, it decomposes to form mercury metal.
(d) Anode - Thick block of impure metal M
Cathode - Thin strip of pure metal M
Electrolyte - Water soluble salt (of metal M).
(e) (i) Copper
(ii) Zinc
(iii) Nickel
(iv) Gold
(v) Silver
Solution 61
(a) Zinc
(b) Cinnabar; Mercury (II) sulphide, HgS
(c) The concentrated copper (I) sulphide ore (copper glance), Cu2S is roasted in air when a part of copper (I) sulphide is oxidised to copper (I) oxide.
When a good amount of copper (I) sulphide has been converted to copper (I) oxide, then the supply of air for roasting is stopped. In the absence of air, copper (I) oxide formed above reacts with remaining copper (I) sulphide to form copper metal and sulphur dioxide.
(d) An alloy is a homogeneous mixture of two or more metals (or a metal and small amounts of non-metals).
Steel and Brass are examples of alloys.
(e) (i) Alloys are stronger than the metals from which they are made.
(ii) Alloys are harder than the constituent metals.
(iii) Alloys are more resistant to corrosion.
(iv) Alloys have lower melting points than constituent metals.
(v) Alloys have lower electrical conductivity than pure metals.
Metals and Non-metals Exercise 195
Solution 92
(a) A is sodium and B is chlorine
(b) C is sodium chloride and D is sodium hydroxide
(c) C will have no effect on litmus solution since it is neutral in nature.
(d) B is a gas at room temperature.
(e) EB3
Solution 93
(a) Mercury, Hg
(b) Cinnabar, HgS
(c) 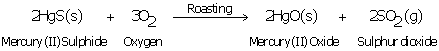
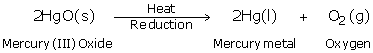
(d) Thermometer
(e) No; Because it is less reactive than copper.
Metals and Non-metals Exercise 196
Solution 94
(a) A is iron (III) oxide and B is aluminium powder.
(b) C is molten iron metal and D is aluminium oxide.
(c)
(d) This reaction is called thermite reaction. It is used for welding of broken pieces of heavy iron objects like railway tracks, etc.
(e) Displacement reactions and oxidation-reduction reactions.
Solution 95
(a) Positively charged carbon electrode (Anode)
(b) This carbon electrode is burnt away because oxygen produced during the electrolysis of molten aluminium oxide reacts gradually with the carbon of carbon anode to form carbon dioxide gas.
Solution 96
(a) X is aluminium and Y is zinc.
(b) X is more reactive than Y.
(c) Bauxite; Al2O3.2H2O
(d) Calamine, ZnCO3
(e) Alloy of metal X : Duralumin ; Alloy of metal Y : Brass
Solution 97
(a) Silver
(b) Copper
(c) Silver sulphide
(d) Basic copper carbonate
(e) Dilute acid solution; The acid solution dissolves green coloured basic copper carbonate present on the corroded copper object makes it look shiny, red brown again.
Solution 98
Metal P is zinc; Metal Q is tin; Metal R is lead; Metal S is iron.
Metal P (zinc) is used to form a thin layer on metal S (iron) by the process of galvanisation to prevent its corrosion.
Metal Q (tin) is used for electroplating tiffin boxes made of metal S (iron).
Metal R (lead) is used in making car batteries.
Metals Q (tin) and R (lead) form an alloy called solder.
Solution 99
(a) Manganese
(b) Manganese dioxide
(c) Aluminium
(d) Chromium
Solution 100
Metal X is gold and Metal Y is silver; The colour of metal X (gold) is yellow.

