ICSE Class 8 Answered
describe the preparation of hydrogen in the laboratory
Asked by nathsantanukumar | 23 Jun, 2018, 20:44: PM
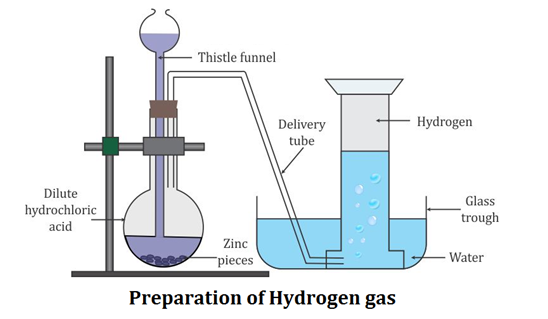
Laboratory Preparation of Hydrogen:
Hydrogen is prepared in the laboratory by the action of the dilute hydrochloric acid or dilute sulphuric acid on granulated zinc.
Use of Granulated Zinc
Granulated zinc contains an impurity like copper which acts as a positive catalyst. A positive catalyst increases the rate of a chemical equation. This is the reason why granulated zinc is preferred over pure zinc for the laboratory preparation of hydrogen gas.
Reaction:
|
Metal + Dilute acid → Salt + Hydrogen
Zn + 2HCl → ZnCl2 + H2 ↑ Zn + H2SO4 → ZnSO4 + H2 ↑ |
Collection of Gas: Hydrogen gas is collected by downward displacement of water.
Answered by Ramandeep | 25 Jun, 2018, 11:25: AM
Concept Videos
ICSE 8 - Chemistry
Asked by vinodlucknow251 | 08 Mar, 2024, 14:27: PM
ICSE 8 - Chemistry
Asked by palashmit53 | 08 Mar, 2022, 18:54: PM
ICSE 8 - Chemistry
Asked by sunitashah0104 | 26 May, 2020, 21:51: PM
ICSE 8 - Chemistry
Asked by SindujaAlluri17 | 09 Jan, 2020, 15:35: PM
ICSE 8 - Chemistry
Asked by Aparajitahatibaruah | 30 Dec, 2019, 20:54: PM
ICSE 8 - Chemistry
Asked by knkprasad5669 | 18 Nov, 2019, 21:43: PM
ICSE 8 - Chemistry
Asked by sn09011977 | 08 Jun, 2019, 09:51: AM
ICSE 8 - Chemistry
Asked by chaudharysn165 | 21 Sep, 2018, 19:57: PM
ICSE 8 - Chemistry
Asked by nathsantanukumar | 23 Jun, 2018, 21:01: PM
ICSE 8 - Chemistry
Asked by nathsantanukumar | 23 Jun, 2018, 21:00: PM