Ethers Preparation And Properties Free Doubts and Solutions
CBSE - XII Science - Chemistry - Alcohols, Phenols and Ethers
pls solve sir
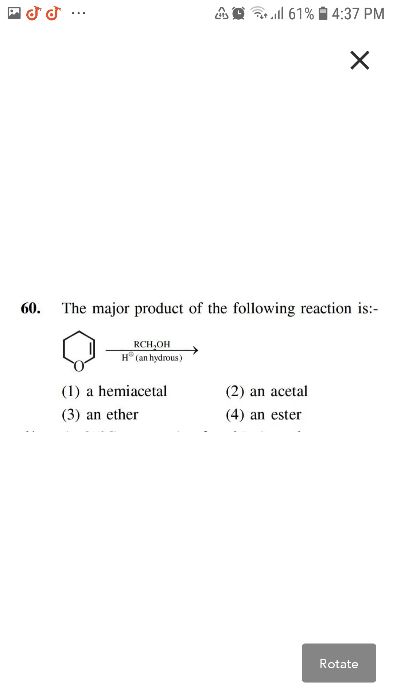
CBSE - XII Science - Chemistry - Alcohols, Phenols and Ethers
Answer 5th with reaction

CBSE - XII Science - Chemistry - Alcohols, Phenols and Ethers
How many other contributing resonance structures are possible?
CBSE - XII Science - Chemistry - Alcohols, Phenols and Ethers
Can you please give me the conversion charts of organic chemistry and previous year questions of every chapters ?
CBSE - XII Science - Chemistry - Alcohols, Phenols and Ethers
actually m solving ncert so i had problem in some question
1. reaction of propanone with methymagnesium bromide followed by hydrolysis
2. hyration of propene in the prensen of dilute sulphuric acid
3 catalytic reduction of butanal
give me in proper explanation its important i want that ans
CBSE - XII Science - Chemistry - Alcohols, Phenols and Ethers
My question is that Single displacement reactions are reactions where one reactant replaces part of the other.
AB + C → AC + B//mechanism
Double displacement reactions are reactions where part of one reactant is replaced by part of another reactant.
AB + CD → AD + CB//mechanism
in inorganic chemistry.
by AB AND CD mechanism we can figure out AC +B is a product of single displacement reaction.
In these type of reactions we can write their products of their reactants.
but in organic chemsitry we cannot write products of the reactants like susbstituion,nucleophilic reaction.Iget confused.
Like in inroganic chemistry we have
AB + C → AC + B//mechanism so are there mechanism in organic chemistry by that twe can write products when reactants are reacting.
AB + C → AC + B//mechanism
Double displacement reactions are reactions where part of one reactant is replaced by part of another reactant.
AB + CD → AD + CB//mechanism
AB + C → AC + B//mechanism so are there mechanism in organic chemistry by that twe can write products when reactants are reacting.
CBSE - XII Science - Chemistry - Alcohols, Phenols and Ethers
How would you account for the following: (i) Phenols are much more acidic than alcohols. (ii) The boiling points of ethers are much lower than those of the alcohols of comparable molar masses.
CBSE - XII Science - Chemistry - Alcohols, Phenols and Ethers
What is the IUPAC name of C6H5OCH3?
CBSE - XII Science - Chemistry - Alcohols, Phenols and Ethers
What is the IUPAC name of the following compound?
CBSE - XII Science - Chemistry - Alcohols, Phenols and Ethers
How ethers are prepared by Williamson's method?
CBSE - XII Science - Chemistry - Alcohols, Phenols and Ethers
How are ethers classified? Give example.
CBSE - XII Science - Chemistry - Alcohols, Phenols and Ethers
On what basis does the R-O Cleavage takes place when R-O-R reacts with HI ?
eg-: CH3-CH2-CH2-O-CH3 + HI ---> CH3-CH2-CH2-OH + CH3I ,
Why is the product not CH3-CHI-CH3 + CH4 ?
eg-: CH3-CH2-CH2-O-CH3 + HI ---> CH3-CH2-CH2-OH + CH3I ,
Why is the product not CH3-CHI-CH3 + CH4 ?

