Could someone explain nucleophilic substitution reaction?
SN1 reactions
SN1 reactions are basically unimolecular nucleophilic substitution reactions. The mechanism of SN1 reactions is as follows.
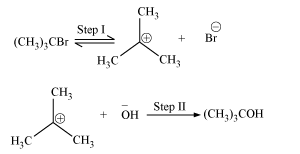
These reactions are carried out in polar protic solvents such as water, alcohol, acetic acid, etc.
The increasing order of reactivity is
1o halide < 2o halide < 3o halide
The reason behind the above trend is as follows.
Greater the stability of a carbocation, more easily the alkyl halide is formed and hence, faster is the reaction rate.
The increasing order of stability of carbocation is 1 o < 2 o < 3o. Since 1 o halide forms 1 o carbocation, 2 o halide forms 2 ocarbocation, and 3 o halide forms 3 o carbocation. Therefore, the increasing order of reactivity is 1 o halide < 2 o halide < 3 ohalide.
SN2 reactions
SN2 reactions are known as bimolecular nucleophilic substitution reactions. The mechanism for these types of reaction is as follows.
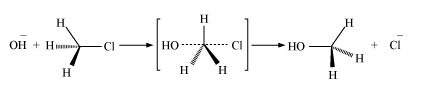
In these reactions, inversion of configuration takes place. The increasing order of reactivity is
3 o halide < 2 o halide < 1 o halide
The reason behind the above trend is as follows.
Due to the presence of bulky substituents in 3 o and 2 ohalides, nucleophile cannot approach.
For both SN1 and SN2 reaction, the order of reactivity of halides is
R−F << R−Cl < R−Br < R−I
The stereochemical aspects of nucleophilic substitution recation can be summarized as
In SN1 reaction, racemisation takes place and thus, follow retention of configuration.
In SN2 reaction, complete stereochemical inversion of configuration takes place.
- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- 9
- 10
You have rated this answer /10
Browse free questions and answers by Chapters
- 1 Classification of Elements and Periodicity in Properties
- 2 Chemical Bonding and Molecular Structure
- 3 States of Matter
- 4 Equilibrium
- 5 Hydrogen
- 6 Hydrocarbons
- 7 Environmental Chemistry
- 8 Solutions
- 9 Chemical Kinetics
- 10 Surface Chemistry
- 11 Biomolecules
- 12 Polymers
- 13 Chemistry in Everyday Life
- 14 Atomic Structure
- 15 Chemical Thermodynamics
- 16 Redox Reactions and Electrochemistry
- 17 p-Block Elements
- 18 d - and f - Block Elements
- 19 Some Basic Principles of Organic Chemistry
- 20 Organic Compounds Containing Halogens
- 21 Organic Compounds Containing Oxygen
- 22 Organic Compounds Containing Nitrogen
- 23 Co-ordination Compounds
- 24 Purification and Characterisation of Organic Compounds
- 25 s-Block Element (Alkali and Alkaline Earth Metals)
- 26 Solid State
- 27 Some Basic Concepts in Chemistry
- 28 General Principles and Processes of Isolation of Metals
- 29 Principles Related to Practical Chemistry
