A copper concentrate (CuFeS2 + FeS2 + gangue) of composition (wt%) Cu = 6%, S = 35% is
roasted with (20+RN*)% excess air. Copper in the roasted product is as Cu2S and all the iron is
converted to Fe2O3. Assuming that the gangue does not contain any Cu, Fe and S, Calculate for 1
ton of concentrate i) amount of minerals and gangue ii) volume of air supplied for roasting at STP.
iii) Volume and composition of the evolved gas (flue) after roasting [atomic weight of Cu=64, Fe=
56, S=32, O=16, N=14]
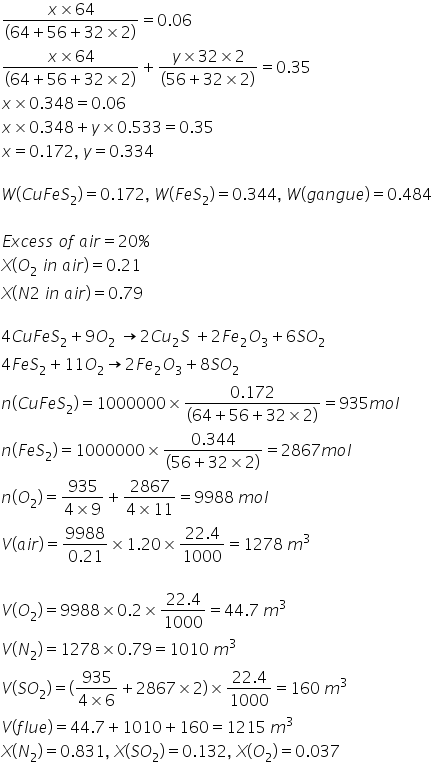
- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- 9
- 10
You have rated this answer /10
- Biology Question Answers for ICSE Class 12 Science
- Chemistry Question Answers for ICSE Class 12 Science
- Economics Question Answers for ICSE Class 12 Science
- Hindi Question Answers for ICSE Class 12 Science
- Maths Question Answers for ICSE Class 12 Science
- Physics Question Answers for ICSE Class 12 Science
Browse free questions and answers by Chapters
- 1 The Solid State
- 2 Solutions
- 3 Electrochemistry
- 4 Chemical Kinetics
- 5 Surface Chemistry
- 6 Isolation of Elements
- 7 The p-Block Elements
- 8 The d-Block and f-Block Elements
- 9 Coordination Compounds
- 10 Haloalkanes and Haloarenes
- 11 Alcohols, Phenols and Ethers
- 12 Aldehydes, Ketones and Carboxylic Acids
- 13 Amines
- 14 Biomolecules
- 15 Polymers
- 16 Chemistry in Everyday Life
