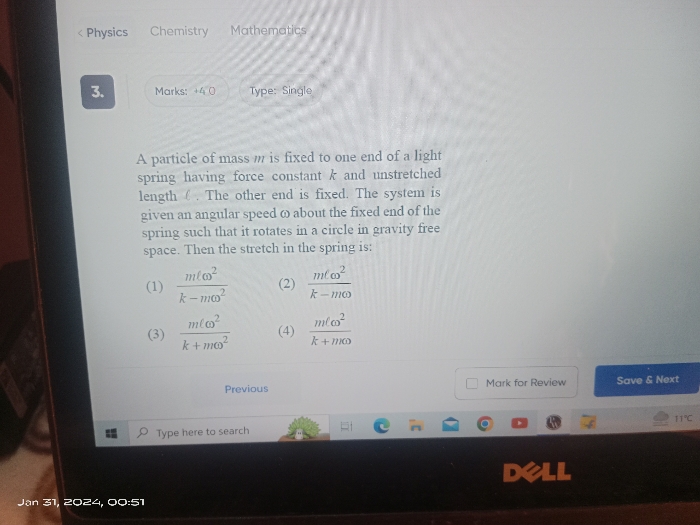
Properties of Solids and Liquids
Properties of Solids and Liquids PDF Notes, Important Questions and Synopsis
SYNOPSIS
- Deforming force: A deforming force is the one which applied changes the configuration of the body.
- A perfectly elastic body immediately and completely regains its original configuration after removal of a deforming force, whereas a body which does not gain its original shape and size after removal of a deforming force, howsoever small the deforming force may be, is called a perfectly plastic body.
- Stress is the restoring force per unit area, and strain is the fractional change in dimension.
- Types of stresses: Tensile stress/longitudinal stress, shearing stress and hydraulic stress.
- Hooke’s law: The extension is proportional to the force or tension in a wire if the proportional limit is not exceeded. The constant of proportionality is called the modulus of elasticity.
- Three elastic moduli—Young’s modulus, shear modulus and bulk modulus—are used to describe the elastic behaviour of objects as they respond to deforming forces which act on them.
- The reciprocal of bulk modulus of elasticity is called compressibility. Bulk modulus of a solid is about 50 times that of a liquid, and bulk modulus of a gas is 10-3 times that of a solid.
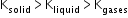
- Strain is fractional deformation.
- Stress is proportional to strain. The proportionality constant is called the elastic modulus.

- Elastomers, a new class of solid, do not obey Hooke’s law.
- Tensile stress = force per unit area = F/A
- Tensile strain is extension per unit length = e/l
- Shearing stress is possible only in solids.
- The material which stretches to a lesser extent for a given load is considered more elastic.
- Young’s modulus and shear modulus are relevant only for solids since only solids have lengths and shape.
- Metals have a larger value of Young’s modulus than alloys and elastomers. A material with a large value of Young’s modulus requires a large force to produce a small change in its length.
- In elastic behaviour, a metal returns to its original length after the load is removed. In this case, the energy is then recovered.
- Stress is not a vector quantity since, unlike force, stress cannot be assigned a specific direction.
- In plastic behaviour, a metal is permanently strained after the load is removed. In this case, the energy is transferred to heat after the elastic limit is exceeded.
Diagrams
Stress–strain curve for a metal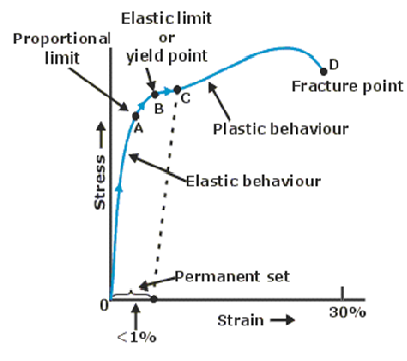
- A liquid is incompressible and has a free surface of its own. A gas is compressible and it expands to occupy all the space available to it.
- Liquids and gases together are known as fluids.
- Pressure at a point is force per area, and it is a scalar quantity. The unit of pressure is pascal (Pa).is defined as the ratio of the force to area.
Its SI unit is N m−2. - Average pressure Pav is defined as the ratio of the force to area.
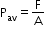
- Pascal, the unit of pressure, is the same as N m−2. Other common units of pressure are
1 atm = 1.01 × 105 Pa
1 bar = 105Pa
1 torr = 133 Pa = 0.133 kPa
1 mm of Hg = 1 torr = 133 Pa - Pressure is defined as normal force per unit area.
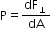
- The pressure difference between two points in a static fluid of uniform density r is proportional to the depth h.
- Pascal’s law: A change in pressure at any point in an enclosed fluid at rest is transmitted undiminished to all points in the fluid.
- Gauge pressure measures the excess pressure above the atmospheric pressure.
- Flow of a fluid whose density is independent of both position and time is said to be incompressible.
- If the frictional forces in a moving fluid are negligible, then the flow is called non-viscous.
- If a fluid element has a non-zero angular velocity at every point, then the flow is said to be non-rotational.
- Orderly flow of a fluid is called streamlined or steady flow.
- In streamlined flow, every liquid element crossing a point has the same velocity.
- Disorderly flow of a fluid is called turbulent flow.
- A streamline is defined as a curve such that the tangent to any point on the curve gives the direction of fluid flow at that point.
- In a steady or streamline flow, no two streamlines ever cross each other.
- The greater the spacing between streamlines in a region, the smaller is the fluid velocity there.
- A bundle of streamlines forming a tubular region is called a tube of flow.
- When the flow is incompressible, non-viscous, steady and non-rotational, it is called ideal fluid flow.
- Equation of continuity: The product of area of cross section and velocity remains constant throughout the flow.
- In case of varying density or compressible liquids, the equation of continuity modifies to product of the density, area of cross section and velocity of the flow remaining constant as opposed to Av = constant.
- If the fluid velocity is less than a certain limiting value called critical velocity, then the flow is steady or streamlined. As the speed of the fluid exceeds the critical velocity, it becomes turbulent.
- Equation of continuity tells us that fluid speed is greater in narrow regions as compared to wider regions.
- If the speed of a fluid element increases as it flows, then the pressure of the fluid must decrease and vice versa. This is one implication of Bernoulli’s principle.
- Bernoulli was the first one to relate this pressure difference to velocity changes.
- Bernoulli also explained the relation between the height of a fluid and changes in pressure and speed of the fluid.
- Bernoulli’s principle: Along a streamline, the sum of the pressure, the kinetic energy per unit volume and the potential energy per unit volume remains constant.
- Bernoulli’s principle holds true in the case of ideal fluid flow, which is incompressible, irrotational and streamlined.
- Bernoulli's principle, which results from conservation of energy, relates the height, pressure and speed of an ideal fluid whether it is a liquid or a gas.
- The speed of outflow of a liquid from a hole in an open tank is called the speed of efflux.
- Velocity of a freely falling fluid is v=
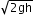 This is called Torricelli’s law.
This is called Torricelli’s law. - A Venturi meter is a device used to measure the flow speed of an incompressible liquid.
- According to Bernoulli’s principle, the pressure above the wing is lower than the pressure below it because the air moves faster above the wing. This higher pressure at the bottom compared to the top applies an upward force to the wing to lift it upwards. This is called dynamic lift.
- Velocity of efflux: Velocity of escaping liquid relative to the container.
- Magnus effect is the curving in the path of the ball introduced due to the difference in pressure above and below the ball.
- The speed of efflux from a hole in an open tank is given by Ö2gh.
- An ideal fluid is incompressible and non-viscous.
- Viscosity describes a fluid's internal resistance to flow and may be thought of as a measure of fluid friction.
- A viscous fluid flows fastest at the centre of the cylindrical pipe and is at rest at the surface of the cylinder.
- Viscosity is internal friction in a fluid.
- Surface tension is due to molecular forces.
- The difference in energy of bulk molecules and surface molecules gives rise to surface tension.
- Drops have a spherical shape because a spherical shape has the minimum surface area for a given volume of a free liquid.
- Surface tension is also responsible for the wiggling of soap bubbles.
Greater the attractive force between molecules of a liquid, greater is its surface tension and greater is its resistance to an increase in the surface area. - Surface tension can be quantitatively defined as the energy required per unit increase in surface area.
- The angle of contact is the angle formed between the solid/liquid interface and the liquid/vapour interface. A vertex is formed where the three interfaces meet.
- When the contact angle is acute, the liquid wets the solid. Example: Water on a glass surface.
- When the contact angle is obtuse, the liquid does not wet the solid. Example: Water on flower petals.
- The surface tension of a liquid decreases with the rise in temperature because molecules get extra energy to overcome their mutual attraction.
- Due to surface tension, the liquid surface squeezes itself to minimum surface area.
- The greater the surface tension of the liquid, greater is the excess pressure required for bubble formation inside it.
- Capillary action is the tendency of a liquid to rise in narrow tubes due to surface tension.
The height of a liquid column rising in a capillary tube depends- On its contact angle q
- Directly on its surface tension S
- Inversely on its density r
- Inversely on radius r of the tube
- Addition of detergent in water lowers the surface tension which helps with the cleansing action.
Download complete content for FREE 
JEE Main - Physics
Asked by indiradevi7342764 | 05 Feb, 2024, 06:36: PM
JEE Main - Physics
Asked by abhisheknand5151 | 10 Jan, 2024, 02:13: PM
JEE Main - Physics
Asked by chandanagangireddy2007 | 19 Dec, 2023, 02:41: PM
JEE Main - Physics
Asked by madasutarun217 | 27 Nov, 2023, 08:25: PM
JEE Main - Physics
Asked by kandurivarshithreddy2010 | 02 Nov, 2023, 08:23: PM
JEE Main - Physics
Asked by jangirayan10 | 10 Jul, 2022, 11:23: AM
Related Chapters
- Physics and Measurement
- Kinematics
- Laws of Motion
- Work, Energy and Power
- Rotational Motion
- Gravitation
- Thermodynamics
- Kinetic Theory of Gases
- Oscillations and Waves
- Electrostatics
- Current Electricity
- Magnetic Effects of Current and Magnetism
- Electromagnetic Induction and Alternating Currents
- Electromagnetic Waves
- Optics
- Dual Nature of Matter and Radiation
- Atoms and Nuclei
- Electronic Devices
- Communication Systems