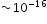Physics and Measurement
Physics and Measurement PDF Notes, Important Questions and Synopsis
SYNOPSIS
- Physics domain:
- Macroscopic domain: This includes phenomena at the laboratory, terrestial and astronomical scales. Classical mechanics deals with the macroscopic domain.
- Microscopic domain: This includes atomic, molecular and nuclear phenomena. Quantum mechanics deals with the microscopic domain.
- Four fundamental forces of nature:
|
Force |
Relative strength |
Range |
|
Gravitational force |
10-39 |
Infinite |
|
Nuclear force (weak) |
10-13 |
Very short, sub-nuclear size |
|
Electromagnetic force |
10-2 |
Infinite |
|
Nuclear force (strong) |
1 |
Short, nuclear size |
- Physical quantities:
-
Measure of physical quantity (Q) = numerical value of physical quantity (n) × size of unit (u) n1u1 = n2u2 = constant
-
Fundamental quantities, SI unit and their symbols:
Fundamental quantity
SI unit
Symbol
Length
metre
m
Mass
kilogram
kg
Time
second
s
Electric current
ampere
A
Temperature
kelvin
K
Amount of substance
mole
mol
Luminous intensity
candela
cd
- Conversion of units:
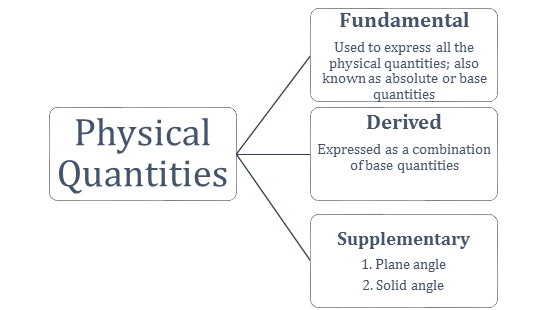
Consider dimensions of a physical quantity as ‘x’ in mass, ‘y’ in length and ‘c’ in time. If the fundamental units in one system are M1, L1 and T1 and in another system are M2, L2 and T2,
then
- Analysis of dimensional correctness:
Every equation should be dimensionally balanced. This is called ‘principle of homogeneity’. The dimensions of each term must be the same on both sides of the equation. - Principle of homogeneity:
According to the principle of homogeneity, dimensions of all terms in a physical expression should be the same. - Errors:
Errors are classified as (i) systematic errors and (ii) random errors.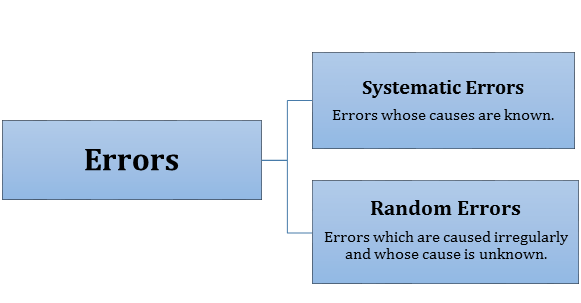
- If x1, x2, x3 … xn are the measured values of quantities in several measurements, then the mean is
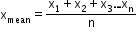
- Absolute errors in measured values are
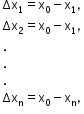
- If the quantity Z is expressed as a product of quantities A and B, then the maximum fractional error in Z is given by
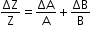
- If Z = AxByCz, then the maximum fractional error in Z is
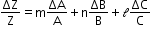
Download complete content for FREE 
JEE Main - Physics
Asked by avhadbalaji2 | 24 Jan, 2024, 02:12: PM
JEE Main - Physics
Asked by kartikchauhan2425 | 13 Jan, 2024, 05:37: PM
JEE Main - Physics
Asked by mvijithareddy67 | 13 Jan, 2024, 09:26: AM
JEE Main - Physics
Asked by amarateja3 | 09 Jan, 2024, 09:26: AM
JEE Main - Physics
Asked by mages032006 | 26 Jun, 2023, 06:37: PM
JEE Main - Physics
Asked by mahadhasyamvarshini | 05 Feb, 2023, 10:25: PM
JEE Main - Physics
Asked by sumchary123 | 12 May, 2022, 04:23: PM
JEE Main - Physics
Asked by shailuupd | 21 Apr, 2022, 09:24: PM
JEE Main - Physics
Asked by harshpandeykp20 | 18 Aug, 2021, 08:31: AM
Related Chapters
- Kinematics
- Laws of Motion
- Work, Energy and Power
- Rotational Motion
- Gravitation
- Properties of Solids and Liquids
- Thermodynamics
- Kinetic Theory of Gases
- Oscillations and Waves
- Electrostatics
- Current Electricity
- Magnetic Effects of Current and Magnetism
- Electromagnetic Induction and Alternating Currents
- Electromagnetic Waves
- Optics
- Dual Nature of Matter and Radiation
- Atoms and Nuclei
- Electronic Devices
- Communication Systems